Crisostomo 2018 MiPschool Tromso C4
| Mitochondrion at the crosstalk between metabolic disease and dysfunction in male fertility? |
Link: MitoEAGLE
Crisostomo L, Rato L, Jarak I, Silva BM, Raposo JF, Oliveira PF, Alves MG (2018)
Event: MiPschool Tromso-Bergen 2018
Male fertility parameters have declined worldwide in recent decades [1], whilst the prevalence of metabolic diseases has increased . Lifestyle, particularly poor food habits linked to high-fat diets, are regarded as a leading cause for the present trend [2], but the biochemical link between diet, metabolic disease and poor male fertility potential is not known yet [3]. Diet reversion (DR) is the first prospective treatment for metabolic disease, but it is not clear to what extent it is effective in recovering male fertility parameters.
In this study we describe the effect of diet reversion in the metabolic and reproductive function of mice. To achieve this, 3 groups of 12 mice were fed with different diets (CTRL – standard mucedola; HFD – high-fat diet and HFDt – high-fat diet for 60 days, then replaced by standard mucedola). Animals were kept in environmental challenging cages for 200 days after weaning, with unrestricted water and food supply. Mice weight, and food and water intake were monitored during the experiment. Metabolic stress tests (IPGTT and IPITT) were performed a week before sacrifice. After sacrifice, several tissues were collected and weighted. Testicular tissue was used for metabolomics analysis by 1H-NMR. Sperm was obtained from epididymis, and mice fertility determined.
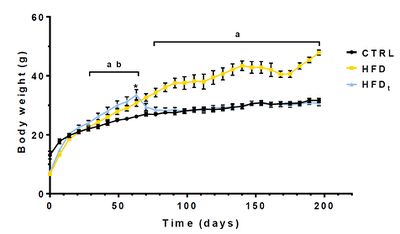
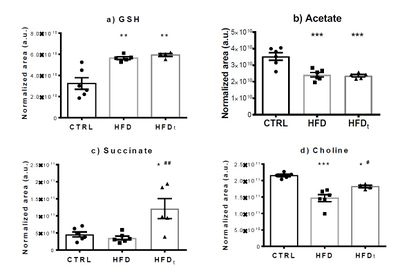
Our results show that DR was effective in recovering the body weight and metabolic function in mice: HFDt mice quickly approximate CTRL mice weight after diet replacement, and both groups perform similarly during the metabolic stress tests. However, HFD and HFDt mice had significantly lower sperm viability and motility than CTRL mice, depicting a limited effect of DR in reproductive function. Metabolomics analysis revealed an altered lipid and energy metabolism in HFD mice, coupled to increased oxidative stress (Figure 2). Notably, HFDt mice presented a significant increase in testicular succinate content, compared to other groups. This accumulation may explain the unique characteristics presented by this group, and elicit a mitochondrial involvement in the changes promoted by DR [4].
Overall our data suggest that high-fat diet compromises male fertility and DR has limited impact on the affected parameters. Thus, weight gain may cause permanent damage on male reproductive tract. Testicular metabolic changes promoted by DR prospect the role of mitochondria as the “missing link” between high-fat diet and male reproductive dysfunction.
• Bioblast editor: Beno M, Plangger M
Affiliations and Support
Crisóstomo L(1,2,3,*), Rato L(4), Jarak I(1,4,5), Silva BM(4), Raposo JF(6,7), Oliveira PF(1,2,3), Alves MG(1)
- Dept Microscopy, Lab Cell Biology, Unit Multidisciplinary Research Biomedicine, Inst Biomedical Sciences Abel Salazar
- Dept Genetics, Fac Medicine
- i3S – Inst Innovation Health Research; Univ Porto, Porto
- Fac Health Sciences, Univ Beira Interior, Covilhã
- Dept Life Sciences, Fac Sciences Technologies Centre Functional Ecology, Univ Coimbra
- NOVA Medical School, Lisbon
- APDP – Associação de Protecção dos Diabéticos de Portugal, Lisbon; Portugal. - [email protected]
This work was supported by the Portuguese Foundation for Science and Technology: Crisóstomo L. (SFRH/BD/128584/2017); M.G. Alves (IFCT2015 and PTDC/BIM-MET/4712/2014); P.F. Oliveira (IFCT2015 and PTDC/BBB-BQB/1368/2014); UMIB (Pest-OE/SAL/UI02015/2014); co-funded by FEDER funds through the POCI/COMPETE 2020; and by the Portuguese Society of Diabetology (SPD) through the “Nuno Castel-Branco” research grant.
Figures
Figure 1: Mice body weight (g) throughout the experiment. Weight of each mice was measured every 7 days. Results are expressed as mean (g) ± SEM (n = 12 for each group). Results were tested by one-way ANOVA with Tukey’s Multicomparison test. Statistical significance was considered when p ≤ 0.05. a) CTRL vs. HFD; b) CTRL vs. HFDt; * Diet Reversion of the HFDt mice.
Figure 2: Absolute quantification of a) GSH – Reduced glutathione, b) acetate, c) succinate and d) choline using 1H-NMR. Results are expressed as mean ± SEM (n = 6 for each condition). Results were tested by one-way ANOVA with Tukey’s Multicomparison test. * Significantly different results relative to control (p < 0.05). # Significantly different results relative to HFD (p < 0.05).
References
- Levine H, Jørgensen N, Martino-Andrade A, Mendiola J, Weksler-Derri D, Mindlis I, Pinotti R, Swan SH (2017) Temporal trends in sperm count: a systematic review and meta-regression analysis. Hum Reprod Update 23:646-59.
- Rato L, Alves MG, Dias TR, Lopes G, Cavaco JE, Socorro S, Oliveira PF (2013) High‐energy diets may induce a pre‐diabetic state altering testicular glycolytic metabolic profile and male reproductive parameters. Andrology 1:495-504.
- Crisóstomo L, Alves MG, Gorga A, Sousa M, Riera MF, Galardo MN, Meroni SB, Oliveira PF (2018) Molecular mechanisms and signalling pathways involved in the nutritional support of spermatogenesis by Sertoli cells, in Sertoli cells - Methods and Protocols. Methods Mol Biol 1748:129-55.
- Tretter L, Patocs A, Chinopoulos C (2016) Succinate, an intermediate in metabolism, signal transduction, ROS, hypoxia, and tumorigenesis. Biochim Biophys Acta 1857:1086-101.
Labels: MiParea: Exercise physiology;nutrition;life style
Organism: Mouse
Tissue;cell: Genital
Event: C4, Oral