Lippe 2015 Abstract MiP2015
| The channel function of the F-ATP synthase complex and its role in the mitochondrial permeability transition. |
Link:
Lippe G, Szabo I, Bernardi P (2015)
Event: MiP2015
Mitochondria are fundamental to cell life and death because they not only supply the bulk of cellular ATP through oxidative phosphorylation, but also have an essential role in free radical signalling and harbour both pro-apoptotic and anti-apoptotic proteins.
In the presence of oxygen, mitochondria operate the exergonic flow of electrons along the respiratory complexes, which is coupled to proton pumping from the matrix to the intermembrane space. The resulting proton motive force drives the backflow of protons through the c-ring in the Fo sector of ATP synthase, leading to the rotation of the F1 γ, δ, and ε subunits within the F1 α3β3 subcomplex, thereby supporting the synthesis of 3 ATP molecules for each 360° rotation. Mitochondria also harbor a regulated channel, the permeability transition pore (PTP), whose radius in mammalian mitochondria is estimated to be about 1.4 nm. PTP opening requires matrix Ca2+ and oxidative stress and is modulated by many effectors including reactive oxygen species, matrix cyclophilin D, Pi, and matrix pH. When PTP opening becomes long-lasting, it causes collapse of the proton gradient preventing ATP synthesis, as well as equilibration of ionic gradients and solutes leading to mitochondria swelling, cristae unfolding, and eventually rupture of the mitochondrial outer membrane accompanied by release of pro-apoptotic proteins [1].
The nature of the PTP has remained a mystery for 60 years until we recently demonstrated that ATP synthase dimers can reversibly undergo a Ca2+-dependent transition to form the PTP [2]. This finding was made possible by 2 sets of critical observations. The first was that CyPD interacts with the ATP synthase at the lateral stalk connecting Fo to F1, and that CyPD interaction is favored by Pi and counteracted by CsA with matching effects on the catalytic activity [3]. The second insight was the identification of subunit oligomycin sensitivity conferring protein (OSCP) as the binding site of CyPD and of the F-ATP synthase inhibitor Bz-423. After the demonstration that Bz-423 is a PTP inducer, we showed that in planar lipid bilayer experiments purified dimers of F-ATP synthase form channels activated by Ca2+, Bz-423, and oxidative stress with currents typical of the PTP [2]. Channel formation by F-ATP synthase has been demonstrated in B. taurus [2], S. cerevisiae [4], and D. melanogaster [1] and appears to be a novel property of the eukaryotic complex.
Labels: MiParea: Respiration, mt-Membrane, mtDNA;mt-genetics
Event: D1, Oral
MiP2015
Affiliations
1-Dept Food Sc, Univ Udine; 2-Dept Biol, Univ Padova; 3-Dept of Biomedical Sc, Univ Padova, Italy. - [email protected]
Abstract continued
These findings demand an assessment of the modifications of ATP synthase that determine the transition of ATP synthase from an energy-conserving into an energy dissipating device. Our working hypothesis is that the channel forms after a conformational change that would follow replacement of Mg2+ with Ca2+ at the catalytic sites located in the β subunits. In mammalian mitochondria, binding of CyPD or Bz-423 to OSCP would increase the accessibility of Ca2+ to the catalytic sites, resulting in onset of the permeability transition. Once the conformational change has occurred, permeation would take place at the interface between dimers (Figure 1), consistent with the inhibiting effect on PTP formation of genetic ablation of the e and g subunits in yeast, which also inhibits ATP synthase dimerization [4].
Figures
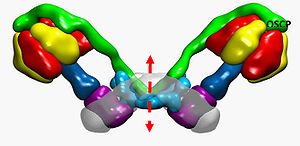
Figure 1. ATP synthase dimers and permeability transition pore formation.
The putative region of channel formation at the interface between dimers (broken arrow) is shown. The F1 α and β subunits are colored in red and yellow, respectively. The F1-rotor γ, δ, and ε subunits are colored in shades of blue, the peripheral stalk subunits b, d, F6, and oligomycin sensitivity conferring protein (OSCP) in shades of green. The position of OSCP is indicated. The c-ring and the remaining FO subunits a, e, f, g, A6L are colored in purple and light blue, respectively. The image (lateral view) has been built starting from the yeast dimer molecular model (PDB ID 4b2q) and superimposing the cryo-electron microscopy map of bovine F-ATP synthase (EMD ID EMD-2091). The molecular model for bovine F-ATP synthase was obtained by superimposing the 3-dimensional structure of the bovine F1-c-ring complex (PDB ID 2xnd) onto each corresponding monomer of the yeast dimer.
References and acknowledgements
- Bernardi P, Di Lisa F, Fogolari F, Lippe G (2015) From ATP to PTP and back: a dual function for the mitochondrial ATP synthase. Circulation Research 116:1850-62.
- Giorgio V, von Stockum S, Antoniel M, Fabbro A, Fogolari F, Forte M, Glick GD, Petronilli V, Zoratti M, Szabó I, Lippe G, Bernardi P (2013) Dimers of mitochondrial ATP synthase form the permeability transition pore. Proceedings Nat Acad Sc U S A 110:5887-92.
- Carraro M, Giorgio V, Šileikytė J, Sartori G, Forte M, Lippe G, Zoratti M, Szabò I, Bernardi P (2014) Channel formation by yeast F-ATP synthase and the role of dimerization in the mitochondrial permeability transition. J Biol Chem 289:15980-5.
- Giorgio V, Bisetto E, Soriano ME, Dabbeni-Sala F, Basso E, Petronilli V, Forte MA, Bernardi P, Lippe G Cyclophilin D modulates mitochondrial F0F1-ATP synthase by interacting with the lateral stalk of the complex. J Biol Chem 284:33982-8.