Vujacic-Mirski 2018 MiPschool Tromso C4
| Development of analytical assays for the detection and quantification of reactive oxygen and nitrogen species in an animal model of type 1 diabetes - ROS formation involving mitochondrial and NADPH oxidase. |
Link: MitoEAGLE
Vujacic-Mirski K, Oelze M, Kroeller-Schoen S, Kalinovic S, Steven S, Muenzel T, Daiber A (2018)
Event: MiPschool Tromso-Bergen 2018
Oxidative stress is a major trigger of endothelial dysfunction and cardiovascular disease [1-3]. Mitochondria and NADPH oxidase contribute to formation of ROS [4,5] and accurate determination of reactive oxygen and nitrogen species (ROS & RNS), especially nitric oxide, superoxide and peroxynitrite, is of great importance for the evaluation of disease mechanisms and the potential targets for drug therapy [6,7]. Our objective is to develop assays to assess mitochondrial, cytosolic and membrane ROS and RNS formation for spatial characterization of the oxidative stress burden in order to identify relevant ROS sources for future therapeutic targeting.
We are working with HPLC methods such as DHE for superoxide production [8-10], Amplex red for hydrogen peroxide detection [8], as well as microplate assays of L-012 chemiluminescence for measuring oxidative burst in in whole blood, cells and tissues [11-14]. Peroxynitrite is indirectly detected by measuring concentration of protein-bound 3-nitrotyrosine (3-NT) by the semi-quantitative method DOT-blot and ELISA kits but also UHPLC with electrochemical detection [15,16], which is the new superior method we are currently working on and could be potentially applied to nitrated mitochondrial proteins as well. MitoSOX-DHE is another method that I currently try to establish in order to detect superoxide production in mitochondria [17]. One of the models of cardiovascular disease we often work with is diabetes, where we use C57Bl/6 mice and streptozotocin (STZ) induced diabetes [18].
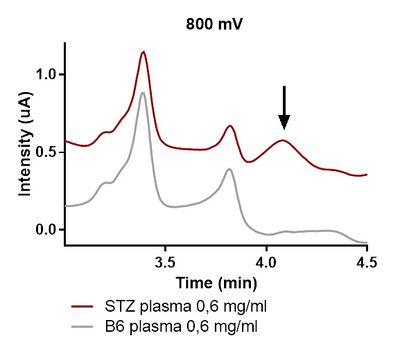
Detection of 3-NT in plasma of control and STZ group clearly shows that the diabetic animals had more 3-NT formation (control < STZ). Although differences can be seen in the chromatographic peaks (Figure 1.), there is a need for optimizing the method for more precise and sensitive quantification of 3-NT. The method of Coularray detection offers high sensitivity and specificity and should be adapted for detecting 3-NT not only in plasma and heart tissues but also in isolated mitochondria and platelets in the future.
• Bioblast editor: Plangger M
Affiliations
- Center Cardiology, Cardiology I, Univ Medical Center Johannes Gutenberg-Univ, Mainz, Germany
Figures
Figure 1. HPLC-Coularray chormatografic profile at 800 mV of mouse plasma of control and STZ treated animals.
References
- Daiber A, Steven S, Weber A, Shuvaev VV, Muzykantov VR, Laher I, Li H, Lamas S, Münzel T (2017) Targeting vascular (endothelial) dysfunction. Br J Pharmacol 174:1591-619.
- Griendling KK, FitzGerald GA (2003) Oxidative stress and cardiovascular injury: Part I: basic mechanisms and in vivo monitoring of ROS. Circulation 108:1912-16.
- Griendling KK, FitzGerald GA (2003) Oxidative stress and cardiovascular injury: Part II: animal and human studies. Circulation 108:2034-40.
- Daiber A (2010) Redox signaling (cross-talk) from and to mitochondria involves mitochondrial pores and reactive oxygen species. Biochim Biophys Acta 1797:897-906.
- Daiber A, Di Lisa F, Oelze M, Kröller-Schön S, Steven S, Schulz E, Münzel T (2017) Crosstalk of mitochondria with NADPH oxidase via reactive oxygen and nitrogen species signalling and its role for vascular function. Br J Pharmacol 174:1670-89.
- Casas AI, Dao VT, Daiber A, Maghzal GJ, Di Lisa F, Kaludercic N, Leach S, Cuadrado A, Jaquet V, Seredenina T, Krause KH, López MG, Stocker R, Ghezzi P, Schmidt HH (2015) Reactive oxygen-related diseases: Therapeutic targets and emerging clinical indications. Antioxid Redox Signal 23:1171-85.
- Schmidt HH, Stocker R, Vollbracht C, Paulsen G, Riley D, Daiber A, Cuadrado A (2015) Antioxidants in translational medicine. Antioxid Redox Signal 23:1130-43.
- Kröller-Schön S, Steven S, Kossmann S, Scholz A, Daub S, Oelze M, Xia N, Hausding M, Mikhed Y, Zinssius E, Mader M, Stamm P, Treiber N, Scharffetter-Kochanek K, Li H, Schulz E, Wenzel P, Münzel T, Daiber A (2014) Molecular mechanisms of the crosstalk between mitochondria and NADPH oxidase through reactive oxygen species-studies in white blood cells and in animal models. Antioxid Redox Signal 20:247-66.
- Kröller-Schön S, Daiber A, Steven S, Oelze M, Frenis K, Kalinovic S, Heimann A, Schmidt FP, Pinto A, Kvandova M, Vujacic-Mirski K, Filippou K, Dudek M, Bosmann M, Klein M, Bopp T, Hahad O, Wild PS, Frauenknecht K, Methner A, Schmidt ER, Rapp S, Mollnau H, Münzel T (2018) Crucial role for Nox2 and sleep deprivation in aircraft noise-induced vascular and cerebral oxidative stress, inflammation, and gene regulation. Eur Heart J doi:10.1093/eurheartj/ehy333.
- Wenzel P, Mollnau H, Oelze M, Schulz E, Wickramanayake JM, Müller J, Schuhmacher S, Hortmann M, Baldus S, Gori T, Brandes RP, Münzel T, Daiber A (2008) First evidence for a crosstalk between mitochondrial and NADPH oxidase-derived reactive oxygen species in nitroglycerin-triggered vascular dysfunction. Antioxid Redox Signal 10:1435-47.
- Daiber A, Oelze M, Steven S, Kroller-Schon S, Munzel T (2017) Taking up the cudgels for the traditional reactive oxygen and nitrogen species detection assays and their use in the cardiovascular system. Redox biology 12:35-49.
- Dikalov SI, Harrison DG (2014) Methods for detection of mitochondrial and cellular reactive oxygen species. Antioxid Redox Signal 20:372-82.
- Debowska K, Debski D, Hardy M, Jakubowska M, Kalyanaraman B, Marcinek A, Michalski R, Michalowski B, Ouari O, Sikora A, Smulik R, Zielonka J (2015) Toward selective detection of reactive oxygen and nitrogen species with the use of fluorogenic probes--Limitations, progress, and perspectives. Pharmacol Rep 67:756-64.
- Zielonka J, Vasquez-Vivar J, Kalyanaraman B (2008) Detection of 2-hydroxyethidium in cellular systems: a unique marker product of superoxide and hydroethidine. Nat Protoc 3:8-21.
- Herce-Pagliai C, Kotecha S, Shuker DE (1998) Analytical methods for 3-nitrotyrosine as a marker of exposure to reactive nitrogen species: a review. Nitric Oxide 2:324-36.
- Daiber A, Daub S, Bachschmid M, Schildknecht S, Oelze M, Steven S, Schmidt P, Megner A, Wada M, Tanabe T, Münzel T, Bottari S, Ullrich V (2013) Protein tyrosine nitration and thiol oxidation by peroxynitrite-strategies to prevent these oxidative modifications. Int J Mol Sci 14:7542-70.
- Zielonka J, Kalyanaraman B (2010) Hydroethidine- and MitoSOX-derived red fluorescence is not a reliable indicator of intracellular superoxide formation: another inconvenient truth. Free Radic Biol Med 48:983-1001.
- Wenzel P, Rossmann H, Müller C, Kossmann S, Oelze M, Schulz A, Arnold N, Simsek C, Lagrange J, Klemz R, Schönfelder T, Brandt M, Karbach SH, Knorr M, Finger S, Neukirch C, Häuser F, Beutel ME, Kröller-Schön S, Schulz E, Schnabel RB, Lackner K, Wild PS, Zeller T, Daiber A, Blankenberg S, Münzel T (2015) Heme oxygenase-1 suppresses a pro-inflammatory phenotype in monocytes and determines endothelial function and arterial hypertension in mice and humans. Eur Heart J 36, 3437-46.
Labels: MiParea: Instruments;methods Pathology: Diabetes Stress:Oxidative stress;RONS Organism: Mouse
Event: C4, Oral