Crisostomo 2019 MiP2019
| “Mitogheist” – Is testicular mitochondrial dysfunction a ghost pattern of paternal high-fat diet in mice? |
Link: MiP2019
Crisostomo L, Rato L, Jarak I, Videira RA, Silva BM, Raposo JF, Oliveira PF, Alves MG (2019)
Event: MiP2019
Despite global efforts to curb the “fat epidemic”, the prevalence of Obesity and related comorbidities, such as diabetes mellitus (DM), is expected to keep its upward trend in the next decades. The temporal trends on sperm parameters [1], have recently raised concern about the burden of metabolic disease in male reproductive potential. High-fat diet (HFD) is regarded as one of the main causes for the “fat epidemics” [2], but it is unclear whether its deleterious effects are inherited by the offspring, by factors carried by the male gamete.
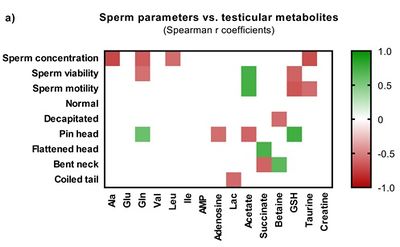
In a recently published study, our group have showed how high-fat diets induce irreversible changes in murine sperm parameters [3], even after diet reversion (DR). Additionally, we have successfully correlated HFD to changes in testicular metabolome which led to anomalies in sperm parameters. Herein we aimed to clarify whether mitochondrial function was affected by HFD and if sperm defects were observed in the offspring.
To pursuit this hypothesis, 3 groups of 12 mice were fed with distinct diets (CTRL – standard mucedola; HFD and HFDt – HFD for 60 days, then replaced by standard mucedola). This population comprised the paternal generation (F0) Animals were kept in environmental challenging cages for 200 days after weaning, with unrestricted water and food supply. Mice weight, and food and water intake were monitored. After sacrifice, several tissues were collected and weighed. Testicular tissue was used for metabolomics analysis by 1H-NMR, and for mitochondrial isolation. Sperm was obtained from epididymis, and mice fertility determined. Western blotting was performed to quantify protein expression of mitochondrial complexes. Individual complexes and citrate synthase activity were further assessed. 120 days post-weaning, males were mated with normoponderal females (1:1) to generate offspring. The same protocols were applied to the resulting offspring generation (F1), but all animals were fed with a standard diet.
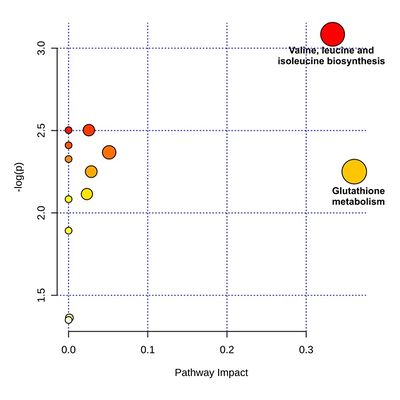
We have previously demonstrated that metabolic pathways directly associated with mitochondrial function were dysregulated in testis of HFD-fed mice, even if transiently. However, no changes were observed on the individual activity of any of the complexes assessed, nor in citrate synthase. Similarly, no changes were observed in complex expression. Strikingly, Complex I activity was significantly decreased in the offspring of HFD comparing to the offspring of CTRL mice. No further changes were observed in the activity nor expression of mitochondrial complexes in the offspring. We were able to trace the global burden of HFD in the offspring testis to branched-chain amino acid metabolism and GSH metabolism, integrating NMR data on MetaboAnalyst 4.0 [4].
Overall our data suggest that HFD has a small impact in the mitochondrial fitness of the testicular tissue, in the diet-challenged animals, but its effects are amplified in the offspring of these mice. Thus, HFD might cause a transgenerational effect in testicular mitochondria, that may be more evident in generations unexposed to HFD (F1 and, possibly, F2 generations). Therefore, paternal HFD leaves a “ghost” signature in mitochondria of the male lineage, that may be expressed in future generations as a pathological phenotype of metabolic dysfunction, without apparent exposure.
• Bioblast editor: Plangger M, Tindle-Solomon L
Labels: MiParea: Developmental biology, Exercise physiology;nutrition;life style
Organism: Mouse
Tissue;cell: Genital
Affiliations and support
- Crisóstomo L(1,2,3), Rato L(4), Jarak I(1,4,5), Videira RA(4), Silva BM(4), Raposo JF(6,7), Oliveira PF(1,2,3), Alves MG(1)
- Dept Microscopy, Lab Cell Biology, Unit Multidisciplinary Research Biomedicine (UMIB), Inst Biomedical Sciences Abel Salazar (ICBAS), Univ Porto, Portugal.
- Dept Genetics, Fac Medicine (FMUP), Univ Porto, Portugal.
- i3S – Inst Innovation Health Research, Univ Porto, Porto, Portugal
- Fac Health Sciences (FCS-UBI), Univ Beira Interior, Covilhã, Portugal
- Dept Life Sciences, Fac Sciences Technologies (FCT-UC) and Centre Functional Ecology (CFE), Univ Coimbra, Portugal
- NOVA Medical School (NMS-NOVA), Lisbon, Portugal
- APDP – Associação de Protecção dos Diabéticos Portugal, Lisbon, Portugal. - [email protected]
- Crisóstomo L(1,2,3), Rato L(4), Jarak I(1,4,5), Videira RA(4), Silva BM(4), Raposo JF(6,7), Oliveira PF(1,2,3), Alves MG(1)
- This work was supported by the Portuguese Foundation for Science and Technology: Crisóstomo L. (SFRH/BD/128584/2017); M.G. Alves (IFCT2015 and PTDC/MEC-AND/28691/2017); P.F. Oliveira (IFCT2015); UMIB (UID/Multi/00215/2019); co-funded by FEDER funds through the POCI/COMPETE 2020; and by the Portuguese Society of Diabetology (SPD) through the GIFT research grant 2019. We acknowledge the Portuguese Nuclear Magnetic Resonance Network (REDE/1517/RMN/2005) for access to their facilities.
Figures
Figure 1: Simplified correlation matrix of Sperm parameters vs testicular metabolites, in the parental (F0) generation (adapted from Crisóstomo et al. 2019). The scale represents Spearman r coefficient values, when p < 0.05 (n = 16, Exact p-value). Data was analysed using GraphPad Prism 8.0.
Figure 2: MetaboAnalyst 4.0 (Chong et al. 2018) Pathway Analysis Tool was used to identify global metabolic burden of paternal HFD in the offspring’s testicular metabolome (F1 generation). A list of metabolites affected by HFD in both parental and filial generations was used as input for the present analysis. The analysis was based on the KEGG database, testing for Inter-betweenness and compared with Fisher’s Exact Test.
References
- Levine H, Jørgensen N, Martino-Andrade A, Mendiola J, Weksler-Derri D, Mindlis I, Pinotti R, Swan SH (2017) Temporal trends in sperm count: a systematic review and meta-regression analysis. Human reprod update 23:646-59.
- Rato L, Alves MG, Dias TR, Lopes G, Cavaco JE, Socorro S, Oliveira PF (2013) High‐energy diets may induce a pre‐diabetic state altering testicular glycolytic metabolic profile and male reproductive parameters. Andrology 1:495-504.
- Crisóstomo L, Rato L, Jarak I, Silva BM, Raposo JF, Batterham RL, Oliveira PF, Alves MG (2019) A switch from high-fat to normal diet does not restore sperm quality but prevents metabolic syndrome. Reproduction.
- Chong J, Soufan O, Li C, Caraus I, Li S, Bourque G, Wishart DS, Xia J (2018) MetaboAnalyst 4.0: towards more transparent and integrative metabolomics analysis. Nucleic Acids Re 46:486-94.