Hickey 2014 Abstract MiP2014
| Mitochondrial inefficiencies and anoxic ATP hydrolysis capacities in diabetic rat heart. |
Link:
Mitochondr Physiol Network 19.13 - MiP2014
Pham T, Loiselle D, Power A, Hickey AJ (2014)
Event: MiP2014
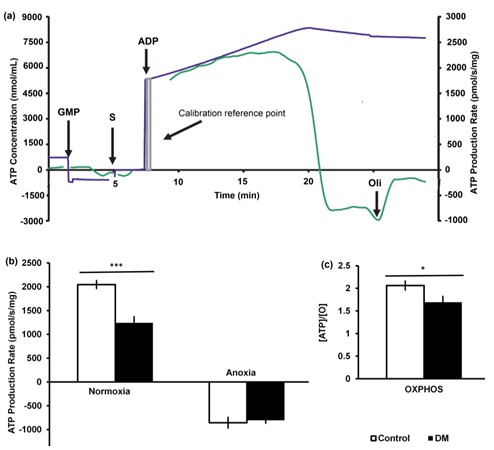
As approximately 80% of diabetics die from heart failure, understanding diabetic cardiomyopathy is crucial. Mitochondria occupy 35-40% of the mammalian cardiomyocyte volume, supply 95% of the hearts’ ATP, and diabetic heart mitochondria show impaired structure, arrangement and function. We predict that bioenergetic inefficiencies are present in diabetic heart mitochondria; therefore, we explored mitochondrial proton and electron handling by linking oxygen flux within streptozotocin (STZ)-induced-diabetic Sprague-Dawley rat heart tissues, to steady-state ATP synthesis, reactive oxygen species (ROS) production and mitochondrial membrane potential (ΔΨ). By coupling high-resolution respirometers with purpose-built fluorometers, we followed Magnesium Green (ATP synthesis), Amplex Ultra Red (ROS production) and safranin-O (ΔΨ). Relative to control rats, the tissue-mass specific respiration of STZ-diabetic hearts was depressed in oxidative phosphorylating (OXPHOS) states. Steady-state ATP synthesis capacity was almost a third lower in STZ-diabetic heart and relative to O2 flux, this equates to an estimated 12% depression in OXPHOS efficiency. However, with anoxic transition, STZ-diabetic and control heart tissues showed similar ATP hydrolysis capacities through reversal of the F1/F0 ATP synthase. STZ-diabetic cardiac mitochondria also produced more net ROS, relative to oxygen flux (ROS/O) in the OXPHOS state. While ΔΨ did not differ between groups, the time to develop ΔΨ with the onset of OXPHOS was protracted in STZ-diabetic mitochondria. ROS/O is higher in life-like OXPHOS states, and potential delays in the time to develop ΔΨ may delay ATP synthesis with inter-beat fluctuations in ADP concentrations. Whereas diabetic cardiac mitochondria produce less ATP in normoxia, they consume as much ATP in anoxic infarct-like states (Fig. 1).
• O2k-Network Lab: NZ Auckland Hickey AJ
Labels: MiParea: Respiration, mt-Membrane Pathology: Cardiovascular, Diabetes, Myopathy Stress:Ischemia-reperfusion, Oxidative stress;RONS Organism: Rat Tissue;cell: Heart Preparation: Permeabilized tissue
Regulation: ATP production, mt-Membrane potential Coupling state: OXPHOS Pathway: N, S HRR: Oxygraph-2k, O2k-Fluorometer Event: B4, Oral MiP2014
Affiliation
1-School Biol Sc Univ Auckland; 2-Fac Medical Health Sc; 3-Auckland Bioengineering Inst, Univ Auckland; New Zealand. - [email protected]
Figure 1
Fig. 1. ATP production capacities (mg-1) of diabetic (black) and age matched control hearts (open). (a) Simultaneous measurement of mitochondrial O2 flux (not shown) and ATP production. Respiratory flux was followed in the presence of oxygen and a CI+II-linked substrate combination (inset a, GMP: glutamate+malate+pyruvate, S: succinate), and then Mg2+-free ADP was added to initiate ATP synthesis (OXPHOS). Respiration was allowed to run into anoxia after which oligomycin (Omy) was added and the background ATP hydrolysis signal subtracted as background. (b) ATP production per mass of tissue (pmol∙s-1∙mg-1) in normoxia and anoxia. (c) The active or steady-state ~P/O ratio was then determined from the rate of ATP synthesised relative to the flux of molecular O in OXPHOS. (*P<0.05, **P<0.01, ***P<0.005, N=12 per group 8-week diabetic rat hearts and their age-matched controls).