Hyunhee 2015 Abstract MiPschool Greenville 2015
| Hypothermic ischemia-normothermic reperfusion-induced necrotic cell death and mitochondrial dysfunction in lung epithelial cells are mediated by RIPK and calpain. |
Link:
Hyunhee K, Keshavjee S, Liu M (2015)
Event: MiPschool Greenville 2015
Lung transplantation is the most effective treatment for patients with end-stage lung diseases. Although the procedure of lung transplantation has significantly improved since its advent in 1983, primary graft dysfunction (PGD) still occurs in 10~20% of post-transplant patients. Ischemia-reperfusion (IR) induced lung injury caused by cold storage condition is one of the major factors of PGD [1]. After 6 h of cold ischemic time (CIT), donor lungs become unsuitable for transplantation, primarily due to tissue deterioration by cell death [2]. Our previous research suggests that necrotic cell death, not apoptotic cell death, is associated with deterioration of post-transplant lung function. In this study, we investigated the intracellular signalling pathways that mediated IR-induced necrosis.
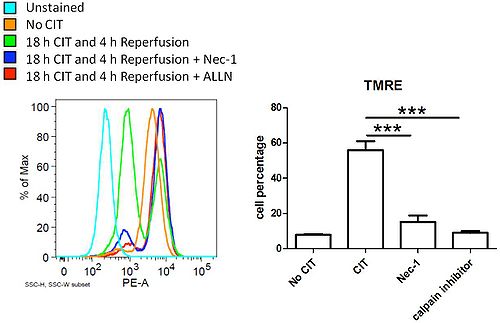
To study IR-induced lung injury at the cellular level, we developed a cell culture model that could simulate lung transplantation. Prolonged CIT (18 h) can induce cell death in donor lungs. To find out whether the cell death is regulated by specific intracellular pathways, various chemical inhibitors known to inhibit necrotic cell death were tested during 18 h CIT and subsequent reperfusion. We further determined the mitochondrial functions by staining cells either with tetramethylrhodamine (TMRE) or mitoSOX followed by flow cytometry.
After 18 h of CIT and 4 h of reperfusion caspase-3 independent cell death was observed in lung epithelial cells. Necrostatin 1 (receptor interacting protein kinase (RIPK) inhibitor) and ALLN (calpain type µ inhibitor) effectively reduced IR-induced cell death in a dose-dependent manner, while cyclosporine A (mitochondrial permeability transition pore inhibitor) and Ca-074 Me (cathepsin B inhibitor) did not. TMRE and mitoSOX experiments revealed that 18 h of CIT and reperfusion decreased mitochondrial membrane potential and increased mitochondrial ROS production, respectively (Fig. 1). The loss of mitochondrial membrane potential and ROS production were inhibited by necrostatin 1 and ALLN.
The findings suggests that prolonged IR can induce regulated necrosis, which appears to be mediated by RIPK and calpain µ, and is associated with mitochondrial dysfunction. Thus, inhibition of RIPK or calpain pathways could potentially be an effective therapy in lung transplantation.
Labels: MiParea: Patients Pathology: Other Stress:Ischemia-reperfusion Organism: Human Tissue;cell: Liver
Event: Poster
Affiliations
1-Latner Thoracic Surgery Research Lab, Toronto General Research Inst, Univ Health Network; 2-Dept Physiology; 3-Dept Surgery, Univ Toronto, Canada. - [email protected]
Figure 1
Figure 1. Necrostatin-1 and ALLN prevented IR induced mitochondrial membrane potential loss
References and acknowledgements
- De Perrot M, Liu M, Waddell TK, Keshavjee S (2003) Ischemia-reperfusion-induced lung injury. Am J Respir Crit Care Med 167:490-511.
- Fischer S, Maclean AA, Liu M, Cardella JA, Slutsky AS, Suga M, Moreira JF, Keshavjee S (2000) Dynamic changes in apoptotic and necrotic cell death correlate with severity of ischemia-reperfusion injury in lung transplantation. Am J Respir Crit Care Med 162:1932-39.