Pereira da Silva Grilo da Silva 2018 MiP2018
| The unspecific effect of etomoxir on mitochondrial respiration. |
Link: MiP2018
Pereira da Silva Grilo da Silva F, Komlodi T, Garcia-Souza LF, Oliveira PJ, Gnaiger E (2018)
Event: MiP2018
The liver mitochondrial dysfunction plays an important role in the progression of non-alcoholic steatohepatitis, therefore improved protocols are required to evaluate mitochondrial fitness in the course of disease. Etomoxir, an inhibitor of carnitine palmitoyltransferase-I, is often used (40-400 μm) in commercial kits to block mitochondrial fatty acid (FA) transport and to modulate respiration during fatty acid oxidation (FAO) [1,2].
To assess the specific effect of Eto on FAO in comparison to unspecific inhibitory side effects on mitochondrial respiration, we tested 40 and 200 μM etomoxir using high-resolution respirometry on permeabilized Huh7 human hepatocellular carcinoma cells and mitochondria isolated from mouse liver and brain mitochondria. We used substrate-uncoupler-inhibitor titration (SUIT) protocols to analyze oxidative phosphorylation (OXPHOS) and electron transfer (ET) capacities of the fatty acid (F) pathway together with the anaplerotic pathways, as well as of the NADH-linked and succinate (S)-pathways separately (F, N, S) and in combination (FN, FS, FNS, NS).
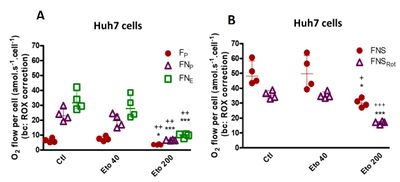
Our results showed that 200 μM etomoxir inhibited not only F-capacity, but also other core mitochondrial metabolic pathways both in liver and brain; however, in brain F-capacity is extremely low compared to liver. 200 μM etomoxir significantly inhibited OXPHOS and ET capacities of N-pathway in Huh7 cells and decreased OXPHOS capacity of N-pathway in isolated liver mitochondria. In contrary, 40 μM etomoxir showed a non-significant inhibitory trend towards N-pathway (Figure 1). After 30 min incubation, 200 μM Eto also blocked ET capacity of FNS-pathway on Huh7 cells (Figure 2B).
Taken together, these results indicate an inhibitory effect of etomoxir on downstream of the Q-junction. The inhibitory pattern observed in the SUIT protocols suggests an unspecific effect of on mitochondrial respiration involving N-and S-pathways and raises caution in its application as specific inhibitor of FAO. Therefore, this work recommends a previous pre-determination of etomoxir toxicity into different models under study before proceeding to use this compound as specific inhibitor of FAO in liver disease models.
• Bioblast editor: Plangger M, Kandolf G
• O2k-Network Lab: AT Innsbruck Oroboros
Affiliations
Silva FSG(1), Komlodi T(2), Garcia-Souza LF(2,3), Oliveira PJ(1), Gnaiger E(2,3)
- Center Neuroscience Cell Biology, Univ Coimbra, Cantanhede, Portugal. - [email protected]
- Oroboros Instruments, Innsbruck
- Dept Visceral, Thoracic Surgery, Medical Univ Innsbruck; Austria
Figures
Figure 1. Effect of etomoxir on NADH-linked (N) pathway, using high-resolution respirometry. The effect of 40 μM and 200 μM etomoxir (Eto) was compared on oxidative phosphorylation capacity (NP, circles) and electron transfer capacity (NE, triangles) of N-pathway to the non-treated samples (Ctl) on permeabilized Huh7 cells (A), mitochondria isolated from liver (B) and brain (C). The results are expressed as median and interquartile range of 4 independent experiments. ***p < 0.001 and **p < 0.01 when compared to the Ctl conditions. +p < 0.05 when compared to 40 μM Eto.
Figure 2. Effect of etomoxir on O2 flux of Huh7 human hepatocellular carcinoma cells after 30 min incubation. The effect of 40 and 200 μM etomoxir (Eto) on oxidative phosphorylation capacity (P) of fatty acid and NADH-linked pathways (FP and FNP, circles and triangles) and the electron transfer capacity (E) (FNE, squares) of 40 μM and 200 μM was compared to the non-treated cells (Ctl) (A). The ET capacity of F-, N- and succinate (S) -pathways in combination are shown with (purple triangles) or without rotenone (red circles) (Rot; FNS, FNSRot) in the absence of Eto (Ctl) or in presence of 40 μM and 200 μM of Eto (B). The results are expressed as median and interquartile range of 4 independent experiments. ***p < 0.001 and *p < 0.05 when compared to the Ctl conditions. +++p < 0.001, ++p < 0.01 and +p < 0.05 when compared to 40 μM Eto.
References and Support
- Clara R, Langhans W, Mansouri A (2016) Oleic acid stimulates glucagon-like peptide-1 release from enteroendocrine cells by modulating cell respiration and glycolysis. Metabolism 65:8-17.
- Kang HM, Ahn SH, Choi P, Ko YA, Han SH, Chinga F, Park AS, Tao J, Sharma K, Pullman J, Bottinger EP, Goldberg IJ, Susztak K (2015) Defective fatty acid oxidation in renal tubular epithelial cells has a key role in kidney fibrosis development. Nat Med 21:37-46.
The mtFOIE GRAS project received funding from the European Union’s Horizon-2020 Research and Innovation program under the Marie Skłodowska-Curie Grant Agreement No.734719. Filomena Silva is recipient of a Post-Doctoral Fellowship from the Foundation for Science and Technology, SFRH/ BPD/122648/2016.
Labels: MiParea: Respiration, Pharmacology;toxicology
Pathology: Cancer, Other
Organism: Human, Mouse Tissue;cell: Nervous system, Liver, Other cell lines Preparation: Permeabilized cells, Isolated mitochondria
Regulation: Inhibitor, Substrate, Uncoupler Coupling state: OXPHOS, ET Pathway: F, N, S, NS, Other combinations HRR: Oxygraph-2k