Rieger 2015 Abstract MiP2015
| pH nanoenvironment sensing in actively respiring mitochondria. |
Link:
Rieger B, Junge W, Busch KB (2015)
Event: MiP2015
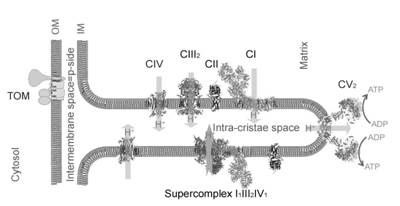
Cristae are flat tube- or disk-like invaginations of the mitochondrial inner membrane extruding into the alkaline matrix space (Figure 1). ATP is mainly produced at the crista membrane by utilization of the proton motive force (pmf = Δψ (membrane potential) + ΔpH) across the membrane. The pmf is the coupling parameter between ATP production and electron transport chain (ETC). Proton pumps and the proton-driven ATP synthase can be spatially segregated. Complexes I-IV are mainly found in the flat sheet membrane of cristae, while Immuno-EM and EM-tomography have revealed ribbons of FoF1 dimers lining the highly curved crista rim [1]. These ribbons seem to be involved in folding the crista membrane. It has been proposed that the concave side of the highly curved rim electrostatically up-concentrates protons at CV to augment the local pmf [2]. By attaching the fluorescent ratiometric pH-sensitive GFP variant pHluorin to OXPHOS complex IV and the dimeric FoF1 ATP synthase, we determined the lateral pH profile along the p-side of cristae in the intra-cristae space (ICS) of living HeLa cells [3]. To stimulate an activated oxidative phosphorylation in glycolytic HeLa cells, glucose was replaced by galactose in the glutamine-containing growth medium [4]. Furthermore, we analysed the effect of metabolic adaptation to the galactose-medium on pH from surface to the bulk of the ICS and correlated it to the matrix pH, Δψ and basal respiration.
Most interesting, we observed that the local pH at FoF1 dimers (proton sink) is by 0.3 units less acidic than at CIV (proton source) in these cells. This finding is consistent with the calculated pH profile for steady proton diffusion from a proton pump in the crista sheet to FoF1 as proton consumer at the rim.
The observed lateral variation in the proton-motive force necessitates a modification to Peter Mitchell’s chemiosmotic proposal. The experimental technique can be extended to other pH-dependent reactions in membrane microcompartments.
Labels: MiParea: mt-Structure;fission;fusion, mt-Membrane
Organism: Human
Tissue;cell: HeLa
Enzyme: Complex IV;cytochrome c oxidase
Event: B1, Oral
MiP2015
Affiliations
Univ Osnabrueck, Dept Biol, Group Mitoch Dynamics. - bettina.rieger@biologie.uni-osnabrueck.de
Figure 1
Figure 1. pH determination in different mitochondrial subcompartments.
References and acknowledgements
- Davies KM, Strauss M, Daum B, Kief JH, Osiewacz HD, Rycovska A, Zickermann V, Kühlbrandt W (2011) Macromolecular organization of ATP synthase and complex I in whole mitochondria. Proc Natl Acad Sci U.S.A 108:14121-26.
- Strauss M, Hofhaus G, Schröder RR, Kühlbrandt W (2008) Dimer ribbons of ATP synthase shape the inner mitochondrial membrane. EMBO J 27:1154-60.
- Rieger B, Junge W, Busch KB (2014) Lateral pH gradient between OXPHOS complex IV and F0F1 ATP-synthase in folded mitochondrial membranes. Nature comm 5.
- Domenis R, Bisetto E, Rossi D, Comelli M, Mavelli I (2012) Glucose-modulated mitochondria adaptation in tumor cells: a focus on ATP synthase and inhibitor factor 1. Int J Mol Sci 13:1933-50.