Siewiera 2017 MiPschool Obergurgl
| Blood platelets as bioenergetic markers of brain damage in STZ-diabetes. |
Link: MitoEAGLE
Siewiera K, Labieniec-Watala M, Wolska N, Polak D, Kassassir H, Watala C (2017)
Event: MiPschool Obergurgl 2017
Nowadays, it is clear to us that many diseases that present clinical symptoms mainly in specific organs can also modulate function (including mitochondrial energetics) of other cells. Translational research is looking for easily accessible cells that can act as sensors of the changes associated with various pathological states, like diabetes, cardiovascular or neurodegeneration diseases. Blood is an excellent source of cells that can be used to monitor changes in different cells and tissues. The use of blood cells has several advantages: a) the collection is easy to carry out and less invasive then a collection of other tissues, b) there is a large number of cells in blood that are easy to isolate, c) blood circulating in the bloodstream is in contact with all other tissues of an organism. For the above mentioned reasons, blood platelets seem to be perfect objects to use as sensors of developing disease and to observe effects of their treatment. Therefore, our aim was to evaluate usefulness of blood platelets as bioenergetic biomarkers of changes in brain mitochondria in diabetic state.
For this purpose twenty 8-week-old Sprague–Dawley rats (males, 180–300 g) were randomly allocated into two groups: healthy, non-diabetic animals (10 rats) and diabetic animals (10 rats). In order to induce diabetes, the animals were intraperitoneally injected with streptozocin (STZ) at the dose of 65 mg/kg of body weight. The control group of animals (healthy) received a vehicle (citrate buffer). Animals, in which blood glucose level was higher than 300 mg/dl after 7 days upon the STZ injection, were considered diabetic and included in the studied group of diabetic animals. After 1 month of confirmed diabetes, the surviving individuals from both groups of animals were anaesthetised with ketamine (100 mg/kg b.w.) and xylazine (23.32 mg/kg b.w.) and the blood was collected from the abdominal aorta into a tube containing 3.2% sodium citrate. Immediately after the blood collection, post-mortem brains were removed and placed in ice-cold MiR05 buffer. In order to isolate blood platelets, the blood was centrifuged (190 g, 12 min, 37 °C, in the presence of 62.5 ng/ml prostaglandin E1) to obtain platelet-rich plasma (PRP). PRP was then centrifuged (1000g, 15 min, 37 °C), the platelet pellet washed in Tyrode's buffer (134 mM NaCl, 0.34 mM Na2HPO4, 2.9 mM KCl, 12 mM NaHCO3, 20 mM HEPES, pH 7.0, 5 mM glucose, 0.35% w/v BSA) with 3.2% sodium citrate (9:1, vol:vol) and centrifuged again in the same conditions. Platelets were resuspended in Tyrode's buffer to achieve a suspension of 1 × 108 platelets/ml. Initially minced brain tissue was transferred to a pre-cooled tubes and homogenized with the use electric homogenizer at low speed on ice. The resulting homogenate was then kept on ice and used for respirometry without further processing. The mitochondrial respiration parameters in intact blood platelets were monitored with the use of high-resolution respirometry (HRR) with a Clark electrode (Oroboros O2k, Oroboros Instruments, Innsbruck, Austria), according to the protocol described by Gnaiger and Renner-Sattler [1], and the brain tissue homogenats were measured according to Benani et al. [2] protocol with a small modification (FCCP titration).
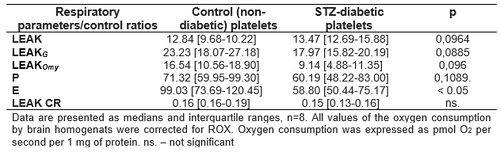
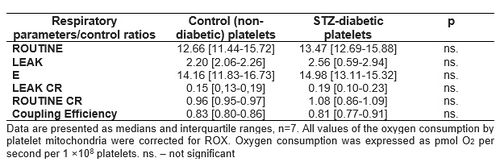
Diabetic animals demonstrated decreased mitochondrial respiration in E state (statistically significant) and reduced respiration (but not statistically significant) without addiction of substrates (LEAK), after glutamate addition (LEAKG) and after oligomycin addition (LEAKOmy) (Table 1). These changes indicate adverse changes in the functioning of the brain mitochondria in STZ-diabetes. However, we did not confirm these changes in the blood platelets, where no changes in respiration between 1 mo.-diabetic and control animals were observed (Table 2).
Our results indicate that in streptozotocin-diabetes model blood platelets are not the best markers of the changes in the brain mitochondria. However, further studies with the use different models of laboratory diabetes and/or animals with longer lasting diabetes are certainly worth of being studied.
• Bioblast editor: Kandolf G
• O2k-Network Lab: PL Lodz Watala C
Labels: MiParea: Respiration Pathology: Diabetes
Organism: Rat Tissue;cell: Blood cells, Platelet Preparation: Intact cells
Coupling state: LEAK, ROUTINE, ET
HRR: Oxygraph-2k Event: C1, Oral
Affiliations
- Siewiera K(1), Labieniec-Watala M(2), Wolska N(1), Polak D(1), Kassassir H(1), Watala C(1)
- 1Dept Haemostasis Haemostatic Disorders, Chair Biomedical Sciences, Medical Univ Lodz
- Fac Biol Environmental Protection, Dept Medical Biophysics, Univ of Lodz,
- Lodz, Poland.- [email protected]
Tables
Table 1. Oxygen consumption and respiratory control ratios calculated for brain tissue homogenates from STZ-diabetic and non-diabetic rats.
Table 2. Oxygen consumption and respiratory control ratios calculated for platelet mitochondria from STZ-diabetic and non-diabetic rats.
References and acknowledgements
- Gnaiger E, Renner-Sattler K. (2009) High-resolution respirometry with cultured cells: a demonstration experiment, In: Gnaiger, E. (Ed.) Mitochondrial Pathways and Respiratory Control. 2nd. Oroboros MiPNet Publications, Innsbruck, pp. 62–73
- Benani A, Barquissau V, Carneiro L, Salin B, Colombani A-L, Leloup C, Casteilla L, Rigoulet M, Pénicaud L (2009) Method for functional study of mitochondria in rat hypothalamus. J Neurosc Meth 178:301–7.
- This study is supported by the National Centre of Science (NCN 2015/17/N/NZ5/02603 and NCN 2016/20/T/NZ3/00505).