Difference between revisions of "Glutamate"
(replaced FW by M and added units g*mol-1) |
(deleted empty line) |
||
| Line 18: | Line 18: | ||
::::# Add 4 mL H<sub>2</sub>O. | ::::# Add 4 mL H<sub>2</sub>O. | ||
::::# Check pH and adjust to 7.0 if necessary with 5 M KOH (usually the pH is 7 without any adjustment); | ::::# Check pH and adjust to 7.0 if necessary with 5 M KOH (usually the pH is 7 without any adjustment); | ||
::::# Adjust final volume to 5 mL and divide into 0.5 mL portions. | ::::# Adjust final volume to 5 mL and divide into 0.5 mL portions. | ||
::::# Store at -20 °C. | ::::# Store at -20 °C. | ||
Revision as of 10:45, 22 March 2021
Description
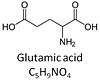
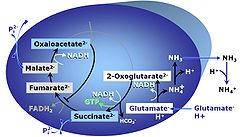
Glutamic acid, C5H9NO4, is an amino acid which occurs under physiological conditions mainly as the anion glutamate-, G, with pKa1 = 2.1, pKa2 = 4.07 and pKa3 = 9.47. Glutamate&malate is a substrate combination supporting an N-linked pathway control state, when glutamate is transported into the mt-matrix via the glutamate-aspartate carrier and reacts with oxaloacetate in the transaminase reaction to form aspartate and oxoglutarate. Glutamate as the sole substrate is transported by the electroneutral glutamate-/OH- exchanger, and is oxidized in the mitochondrial matrix by glutamate dehydrogenase to α-ketoglutarate ( 2-oxoglutarate), representing the glutamate-anaplerotic pathway control state. Ammonia (the byproduct of the reaction) passes freely through the mitochondrial membrane.
Abbreviation: G
Reference: Gnaiger 2020 BEC MitoPathways
Application in HRR
- G: Glutamate (L-Glutamic acid, monosodium salt hydrate, C5H8NO4Na; contains 1 mol H2O /mol); Sigma G 1626, 100 g, store at R.T.; M = 169.1 g·mol-1)
- Preparation of 2 M stock solution (dissolved in H2O)
- Weigh 1.691 g L-Glutamic acid, monosodium salt hydrate, and transfer to a 5 mL volumetric glass flask.
- Add 4 mL H2O.
- Check pH and adjust to 7.0 if necessary with 5 M KOH (usually the pH is 7 without any adjustment);
- Adjust final volume to 5 mL and divide into 0.5 mL portions.
- Store at -20 °C.
- » O2k manual titrations MiPNet09.12 O2k-Titrations
- Titration volume (2-mL O2k-chamber): 10 µL using a 25 µL Hamilton syringe.
- Titration volume (0.5-mL O2k-chamber): 2.5 µL using a 10 µL Hamilton syringe
- Final concentration: 10 mM.
MitoPedia topics: Substrate and metabolite