Mracek 2015 Abstract MiP2015
| Myocardial iron and mitochondrial function in failing and non-failing human heart: direct tissue analysis. |
Link:
Melenovsky V, Mracek T, Petrak J, Pecina P, Benes J, Borlaug B, Drahota Z, Pluhacek T, Nuskova H, Kovalcikova J, Kautzner J, Pirk J, Houstek J (2015)
Event: MiP2015
Very little is known about the determinants and consequences of myocardial iron (Fe) level in normal or failing human myocardium. We hypothesized that myocardial Fe deficiency (ID) in heart failure (HF) is associated with impaired mitochondrial function.
LV samples were obtained from 91 consecutive patients undergoing transplantation (HF: LVEF 23±8%, age 53±11y, 83% males, 46% CAD, 24 % diabetes) and from 38 HF-free organ donors (CON: LVEF 57±12%, age 42±15y, 50% males, 14% with diabetes). Abundance of respiratory chain complexes I-V, ROS-protective enzymes and activities of citrate synthase (CS, Krebs cycle) and RC enzymes NADH-cytochrome c oxidoreductase (NCCR), succinate cytochrome c oxidoreductase (SCCR) and cytochrome c oxidase (COX) and tissue respiration (O2 consumption) were measured in homogenates. Total Fe was measured by inductively-coupled mass spectrometry in lyophilised samples.
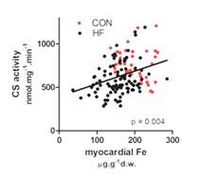
Compared to CON, HF patients had reduced total myocardial Fe (156±41 vs 200±38 µg/g dry weight, p<0.001). Myocardial ID (Fe<124.8 µg/g; 2*SD from the mean of CON) was present in 22 % of HF patients. HF+ID patients had more extensive coronary artery disease, less often betablockers and longer duration of HF, but similar age, gender, renal function, haemoglobin concentration, LVEF or BNP as non-ID HF. Respiratory chain complex I-III, activities of CS, COX, SCCR and NCCR and myocardial tissue respiration were all reduced in HF vs. CON (by 21% - 34%, all p<0.001). In HF, Fe correlated with CS (r=0.30, p=0.004) and SCCR activities (r=0.24, p=0.02), but not with COX or NCCR. Iron-deficient HF patients displayed reduced CS activity (516±145 vs 613±19, p=0.03), reduced abundance of respiratory chain complex III, reduced catalase and glutathione peroxidase.
Myocardial Fe content is systematically reduced in advanced HF and is associated with mitochondrial dysfunction, in particular with diminished CS activity and reduced catalase. These relations may lead to reduced substrate flexibility, decreased energetic production and diminished ROS-defense in iron-deficient failing myocardium.
• O2k-Network Lab: CZ Prague Houstek J, CZ Hradec Kralove Cervinkova Z, CZ Prague Kalous M
Labels: MiParea: Respiration, mt-Medicine, Patients Pathology: Cardiovascular, Diabetes
Organism: Human Tissue;cell: Heart Preparation: Homogenate Enzyme: Complex I, Complex II;succinate dehydrogenase, Complex III, Complex IV;cytochrome c oxidase, Complex V;ATP synthase
Event: A1, Poster
MiP2015
Affiliations
1-Dept Cardiology Cardiac Surgery, Inst Clinical Experimental Med, Prague, Czech Republic; 2-Dept Bioenergetics, Inst Physiology Czech Acad Sc, Prague, Czech Republic; 2-Dept Pathophysiology, 1st Fac Med, Charles Univ, Prague, Czech Republic; 4-Fac Nat Sc, Palackeho Univ, Olomouc, Czech Republic; 5-Dept Cardiovascular Diseases, Mayo Clinic, Rochester, MN, USA. - [email protected]
Figures
Acknowledgements
Research relating to this abstract was funded by Grant Agency of the Ministry of Health of the Czech Republic (NT14050-3/2013).