Difference between revisions of "Baglivo 2022 BEC"
Tindle Lisa (talk | contribs) |
|||
| (4 intermediate revisions by the same user not shown) | |||
| Line 1: | Line 1: | ||
[[File:Bioblast2022 banner.jpg|link=Bioblast_2022]] | [[File:Bioblast2022 banner.jpg|link=Bioblast_2022]] | ||
{{BEC}} | {{BEC}} | ||
{{Publication | {{Publication | ||
|title=Baglivo E, Cardoso LHD, Cecatto C, Gnaiger E (2022) Statistical analysis of instrumental reproducibility as internal quality control in high-resolution respirometry. Bioenerg Commun 2022.8. https://doi.org/10.26124/bec:2022-0008 | |title=Baglivo E, Cardoso LHD, Cecatto C, Gnaiger E (2022) Statistical analysis of instrumental reproducibility as internal quality control in high-resolution respirometry. Bioenerg Commun 2022.8. https://doi.org/10.26124/bec:2022-0008 | ||
|info=Bioenerg Commun 2022. | |info=Bioenerg Commun 2022.08. [[File:OpenAccess-downloadPDF.png|200px||link=https://www.bioenergetics-communications.org/index.php/bec/article/view/baglivo_2022/88 |Open Access pdf]]<br /> ''published online'' 2022-08-04 [[File:WatchThePresentationYoutube_icon.jpg|200px|link=https://www.youtube.com/watch?v=mNSu-hY3hUg&t=15s|»''Watch the presentation''«]]<br /><br /> | ||
<br /><br /> | |||
|authors=Baglivo Eleonora, Cardoso Luiza HD, Cecatto Cristiane, Gnaiger Erich | |authors=Baglivo Eleonora, Cardoso Luiza HD, Cecatto Cristiane, Gnaiger Erich | ||
|year= | |year=2022 | ||
|journal=Bioenerg Commun | |journal=Bioenerg Commun | ||
|abstract=[[File:BEC.png|25px|link=https://doi.org/10.26124/bec:2022-0008]] https://doi.org/10.26124/bec:2022-0008 | |abstract=[[File:BEC.png|25px|link=https://doi.org/10.26124/bec:2022-0008]] https://doi.org/10.26124/bec:2022-0008 | ||
| Line 17: | Line 15: | ||
We applied this SOP in a 3-year study using 48 Oroboros O2k chambers. Stability of air and zero O<sub>2</sub> calibration signals was monitored throughout intervals of up to 8 months without sensor service. Maximum drift over 1 to 3 days was 0.06 pmol∙s<sup>−1</sup>∙mL<sup>−1</sup>, without persistence over time since drift was <0.004 pmol∙s<sup>−1</sup>∙mL<sup>−1</sup> for time intervals of one month, corresponding to a drift per day of 0.2 % of the signal at air saturation. Instrumental O<sub>2</sub> background -d''c''<sub>O<sub>2</sub></sub>/d''t'' was stable within ±1 pmol∙s<sup>−1</sup>∙mL<sup>−1</sup> when measured at monthly intervals. These results confirm the instrumental limit of detection of volume-specific O<sub>2</sub> flux at ±1 pmol∙s<sup>−1</sup>∙mL<sup>−1</sup>. The instrumental SOP applied in the present study contributes to the generally applicable internal quality control management ensuring the unique reproducibility in high-resolution respirometry.<br> | We applied this SOP in a 3-year study using 48 Oroboros O2k chambers. Stability of air and zero O<sub>2</sub> calibration signals was monitored throughout intervals of up to 8 months without sensor service. Maximum drift over 1 to 3 days was 0.06 pmol∙s<sup>−1</sup>∙mL<sup>−1</sup>, without persistence over time since drift was <0.004 pmol∙s<sup>−1</sup>∙mL<sup>−1</sup> for time intervals of one month, corresponding to a drift per day of 0.2 % of the signal at air saturation. Instrumental O<sub>2</sub> background -d''c''<sub>O<sub>2</sub></sub>/d''t'' was stable within ±1 pmol∙s<sup>−1</sup>∙mL<sup>−1</sup> when measured at monthly intervals. These results confirm the instrumental limit of detection of volume-specific O<sub>2</sub> flux at ±1 pmol∙s<sup>−1</sup>∙mL<sup>−1</sup>. The instrumental SOP applied in the present study contributes to the generally applicable internal quality control management ensuring the unique reproducibility in high-resolution respirometry.<br> | ||
|keywords=high-resolution respirometry HRR; polarographic oxygen sensor POS; air calibration; instrumental background; reproducibility; limit of detection; internal quality control IQC; standard operating procedure SOP | |keywords=high-resolution respirometry HRR; polarographic oxygen sensor POS; air calibration; instrumental background; reproducibility; limit of detection; internal quality control IQC; standard operating procedure SOP | ||
|editor=Tindle-Solomon L | |editor=Tindle-Solomon L | ||
|mipnetlab=AT Innsbruck Oroboros | |mipnetlab=AT Innsbruck Oroboros | ||
}} | }} | ||
[[File:BEC-exlibris.png|right|290px|Bioenergetics Communications|link=https://www.bioenergetics-communications.org/index.php/bec/index]] | |||
ORC'''ID''':[[File:ORCID.png|20px|link= https://orcid.org/0000-0002-5507-9663]] Baglivo Eleonora [[File:ORCID.png|20px|link=https://orcid.org/0000-0001-6392-9229]] Cardoso Luiza HD, [[File:ORCID.png|20px|link=https://orcid.org/0000-0001-7068-6165]] Cecatto Cristiane, [[File:ORCID.png|20px|link=https://orcid.org/0000-0003-3647-5895]] Gnaiger Erich | ORC'''ID''':[[File:ORCID.png|20px|link= https://orcid.org/0000-0002-5507-9663]] Baglivo Eleonora [[File:ORCID.png|20px|link=https://orcid.org/0000-0001-6392-9229]] Cardoso Luiza HD, [[File:ORCID.png|20px|link=https://orcid.org/0000-0001-7068-6165]] Cecatto Cristiane, [[File:ORCID.png|20px|link=https://orcid.org/0000-0003-3647-5895]] Gnaiger Erich | ||
__TOC__ | __TOC__ | ||
| Line 34: | Line 32: | ||
|area=Respiration, Instruments;methods | |area=Respiration, Instruments;methods | ||
|tissues=HEK | |tissues=HEK | ||
|instruments=Oxygraph-2k, O2k-Protocol | |instruments=Oxygraph-2k, TIP2k, O2k-Protocol | ||
|additional=BEC, SUIT-001 O2 ce-pce D003 | |additional=BEC, SUIT-001 O2 ce-pce D003 | ||
}} | }} | ||
[[Category:BEC]] | [[Category:BEC]] | ||
Latest revision as of 04:54, 9 March 2023
| Baglivo E, Cardoso LHD, Cecatto C, Gnaiger E (2022) Statistical analysis of instrumental reproducibility as internal quality control in high-resolution respirometry. Bioenerg Commun 2022.8. https://doi.org/10.26124/bec:2022-0008 |
» Bioenerg Commun 2022.08. ![]()
published online 2022-08-04 ![]()
Baglivo Eleonora, Cardoso Luiza HD, Cecatto Cristiane, Gnaiger Erich (2022) Bioenerg Commun
Abstract: ![]() https://doi.org/10.26124/bec:2022-0008
https://doi.org/10.26124/bec:2022-0008
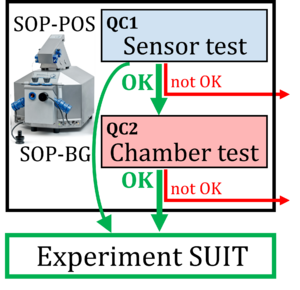
Evaluation of instrumental reproducibility is a primary component of quality control to quantify the precision and limit of detection of analytical procedures. A pre-analytical instrumental standard operating procedure (SOP) is implemented in high-resolution respirometry consisting of: (1) a daily SOP-POS for air calibration of the polarographic oxygen sensor (POS) in terms of oxygen concentration cO2 [µM]. This is part of the sensor test to evaluate POS performance; (2) a monthly SOP-BG starting with the SOP-POS followed by the chamber test quantifying the instrumental O2 background. The chamber test focuses on the slope dcO2/dt [pmol∙s−1∙mL−1] to determine O2 consumption by the POS and O2 backdiffusion into the chamber as a function of cO2 in the absence of sample. Finally, zero O2 calibration completes the sensor test.
We applied this SOP in a 3-year study using 48 Oroboros O2k chambers. Stability of air and zero O2 calibration signals was monitored throughout intervals of up to 8 months without sensor service. Maximum drift over 1 to 3 days was 0.06 pmol∙s−1∙mL−1, without persistence over time since drift was <0.004 pmol∙s−1∙mL−1 for time intervals of one month, corresponding to a drift per day of 0.2 % of the signal at air saturation. Instrumental O2 background -dcO2/dt was stable within ±1 pmol∙s−1∙mL−1 when measured at monthly intervals. These results confirm the instrumental limit of detection of volume-specific O2 flux at ±1 pmol∙s−1∙mL−1. The instrumental SOP applied in the present study contributes to the generally applicable internal quality control management ensuring the unique reproducibility in high-resolution respirometry.
• Keywords: high-resolution respirometry HRR; polarographic oxygen sensor POS; air calibration; instrumental background; reproducibility; limit of detection; internal quality control IQC; standard operating procedure SOP
• Bioblast editor: Tindle-Solomon L
• O2k-Network Lab: AT Innsbruck Oroboros
ORCID:![]() Baglivo Eleonora
Baglivo Eleonora ![]() Cardoso Luiza HD,
Cardoso Luiza HD, ![]() Cecatto Cristiane,
Cecatto Cristiane, ![]() Gnaiger Erich
Gnaiger Erich
Preprint
Support
- This work was part of the Oroboros NextGen-O2k project, with funding from the European Union’s Horizon 2020 research and innovation programme under grant agreement nº 859770.
Labels: MiParea: Respiration, Instruments;methods
Tissue;cell: HEK
HRR: Oxygraph-2k, TIP2k, O2k-Protocol
BEC, SUIT-001 O2 ce-pce D003