| OXPHOS capacity in human muscle tissue and body mass excess – the MitoEAGLE mission towards an integrative database (Version 6; 2020-01-12). |
Link: MiP2019 ![]()
Gnaiger E (2019)
Event: MiP2019
“Preventable diseases are strongly related to a sedentary life style. These are spreading world-wide at an epidemic scale. Mitochondrial dysfunction is increasingly associated with the progression of such pathologies: cause or consequence? There is currently no regimented, quantitative system, or database organized to routinely test, compare and monitor mitochondrial capacities within individuals, populations, or among populations. This reflects the need for scientific innovation and represents a shortcoming in the health system of our modern, rapidly aging society” (MitoEAGLE COST Action application). The working groups of the COST Action CA15203 have made substantial progress towards meeting the mission of Mitochondrial Fitness Mapping (Fig. 1). The present communication (1) provides an example of harmonization of datasets published by different research laboratories on OXPHOS capacity in isolated mitochondria and permeabilized fibers obtained from biopsies of human skeletal muscle (vastus lateralis); (2) emphasizes the importance of comparative protocol harmonization projects and reproducibility studies; (3) illustrates the necessity and difficulty of defining objective exclusion criteria and applying quality assessment of published data; (4) links muscle mitochondrial fitness to whole body aerobic fitness; (5) discusses the extension of tissue-specific to systemic mitochondrial fitness from muscle to brain; and (6) documents the added value of Open Access data repositories.
Analogous to ergometric measurement of VO2max on a cycle or treadmill, cell ergometry is based on measurement of OXPHOS-capacity, JO2,P [pmol O2·s-1·mg-1] equivalent to [µmol O2·s1·kg1], at the mitochondrial level. The main datasets on OXPHOS capacity of isolated mitochondria or permeabilized muscle fibers, harmonization algorithms, and exclusion criteria applied in the present analysis have been reviewed ten years ago [1]. Only a few more studies based on high-resolution respirometry published since then were integrated, exclusively on Caucasian healthy controls [2,3]. This 'MitoEAGLE BME database' is intended to initiate a comprehensive review by the MitoEAGLE Working Group 2 (skeletal muscle). Harmonization introduces potential biases with a scope of improvement based on updated evaluation of (1) wet/dry mass ratios applicable to studies reporting dry mass only; (2) flux control ratios applied to calculate combined NADH- and succinate-linked OXPHOS capacities from data limited to the NADH-pathway or succinate-pathway capacities measured separately; (3) temperature adjustment for measurements at temperatures different from 37 °C [4]; (4) oxygen limitation of measurements with permeabilized fibers that are performed at or below air saturation [5]; (5) OXPHOS capacities reported without evaluation of saturating concentrations of ADP, Pi, and fuel substrates, or without concern of stable steady-state fluxes; and (6) potential bias when results are reported without details on instrumental O2-background tests, calibrations, and corresponding corrections.
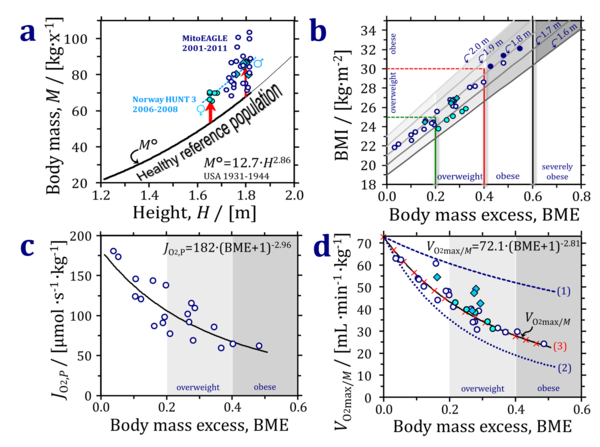
Recent trends of an increasing body mass index (BMI) of the human population indicate an epidemic prevalence of obesity in many countries despite the fact that underweight remains the dominant problem in the world’s poorest regions [6]. Extending the concept of the ‘Reference Man’ [7], a healthy reference population (HRP) is defined with a large range of body height (standing height, H) and corresponding reference body mass, M°, reference V°O2max/M, and mitochondrial fitness parameters (Fig. 2). The reference mass/height relationship constitutes a basic component of the concept of the HRP, obtained from >17.000 measurements on healthy people reported between 1931 and 1944 before the fast food and soft drink epidemic, with about half of the reported measurements ranging from 1.2 to 1.8 m/x corresponding to M° of 22 to 68 kg/x and H/M0.35 [8] (Fig. 2a).
The body mass excess, BME, is defined as the excess of the actual body mass, M-M°, relative to the reference body mass, M°, at the same height (Suppl. Tab. S1). Deviations of M versus M° are due to weight gain without height gain. The similar displacement of men and women (Norwegian HUNT 3 study [9]) from the HRP line is consistent with the increase of average BMI in Norway during the past decades [6]. (Fig. 2a). BME>0 (excess) yields a more consistent index of overweight and obesity across a large range of body heights compared to the BMI (Fig. 2b). Similarly, BME<0 (not shown) indicates a body mass deficit which is insufficiently reflected by the BMI at different body heights. Mitochondrial OXPHOS capacity per mass of vastus lateralis declines as a power function of BME+1=M/M° (Fig. 2c). VO2max/M can be modeled as a function of (1) the metabolically inactive (compared to VO2max) body mass added to a person at height H, (2) the decline of mitochondrial capacity per muscle mass as a consequence of an inactive lifestyle and body mass excess, and (3) a slight increase of muscle mass with increasing BME as a ‘weight lifting effect’ (Fig. 2d).
Taken together, the BME has a strong conceptual foundation on the level of large scale population statistics and is linked to lifestyle and mitochondrial fitness. Importantly, the BME has a straightforward understandable meaning that is easy to communicate to the general public on the personal level: you are overweight if your body mass is increased by 20 % relative to the reference body mass determined by your height. The consequences of mitochondrial control on VO2max/M will be discussed in terms of mechanistic explanations of a large range of neurodegenerative diseases related to the passive lifestyle with an increased BME [10].
Note: (2020-01-12) In the original version of this abstract, BME(old) was defined as M/M°. To be more consistent with the term excess, BME(new) is defined as (M-M°)/M°.
• Keywords: healthy reference population - HRP, body mass index - BMI, body mass excess - BME, aerobic capacity - VO2max per body mass, mitochondrial fitness • Bioblast editor: Plangger M, Tindle-Solomon L, Gnaiger E • O2k-Network Lab: AT Innsbruck Oroboros
Affiliations
- Oroboros Instruments, Innsbruck, Austria
- Dept Visceral, Transplant Thoracic Surgery, Daniel Swarovski Research Lab, Medical Univ Innsbruck, Austria
- Supported by project NextGen-O2k which has received funding from the European Union’s Horizon 2020 research and innovation programme under grant agreement No. 859770. Contribution to COST Action CA15203 MitoEAGLE funded by the Horizon 2020 Framework Programme of the European Union.
References
- Gnaiger E (2009) Capacity of oxidative phosphorylation in human skeletal muscle. New perspectives of mitochondrial physiology. Int J Biochem Cell Biol 41:1837-45. - »Bioblast link«
- Pesta D, Hoppel F, Macek C, Messner H, Faulhaber M, Kobel C, Parson W, Burtscher M, Schocke M, Gnaiger E (2011) Similar qualitative and quantitative changes of mitochondrial respiration following strength and endurance training in normoxia and hypoxia in sedentary humans. Am J Physiol Regul Integr Comp Physiol 301:R1078–87. - »Bioblast link«
- Gnaiger E, Boushel R, Søndergaard H, Munch-Andersen T, Damsgaard R, Hagen C, Díez-Sánchez C, Ara I, Wright-Paradis C, Schrauwen P, Hesselink M, Calbet JAL, Christiansen M, Helge JW, Saltin B (2015) Mitochondrial coupling and capacity of oxidative phosphorylation in skeletal muscle of Inuit and caucasians in the arctic winter. Scand J Med Sci Sports 25 (Suppl 4):126–34. - »Bioblast link«
- Lemieux H, Blier PU, Gnaiger E (2017) Remodeling pathway control of mitochondrial respiratory capacity by temperature in mouse heart: electron flow through the Q-junction in permeabilized fibers. Sci Rep 7:2840. - »Bioblast link«
- Pesta D, Gnaiger E (2012) High-resolution respirometry. OXPHOS protocols for human cells and permeabilized fibres from small biopsies of human muscle. Methods Mol Biol 810:25-58. - »Bioblast link«
- NCD Risk Factor Collaboration (NCD-RisC) (2017) Worldwide trends in body-mass index, underweight, overweight, and obesity from 1975 to 2016: a pooled analysis of 2416 population-based measurement studies in 128·9 million children, adolescents, and adults. Lancet 390:2627–42.
- Sender R, Fuchs S, Milo R (2016) Revised estimates for the number of human and bacteria cells in the body. PLoS Biol 14:e1002533.
- Zucker TF (1962) Regression of standing and sitting weights on body weight: man. In: Altman PL, Dittmer DS, eds: Growth including reproduction and morphological development. Committee on Biological Handbooks, Fed Amer Soc Exp Biol:336-7.
- Loe H, Rognmo Ø, Saltin B, Wisløff U (2013) Aerobic capacity reference data in 3816 healthy men and women 20-90 years. PLoS One 8:e64319.
- Gnaiger E (2020) Body mass excess associated with decline of aerobic capacity and mitochondrial fitness. MitoFit Preprint Arch (in prep).
Figures
Figure 1. Challenges for initiation of a data repository designed to ultimately describe the linkage between the mt-phenotype and anthropometric variables. Modified after MitoEAGLE COST Action application.
- Figure 2: Anthropometrics of a healthy reference population, mitochondrial fitness and aerobic capacity. Full circles and diamonds are the averages of females and males in the 2nd to 6th decade of life with average height of 1.66 and 1.80 m, respectively, from the Norwegian HUNT 3 fitness study [9]. Open circles are averages of the healthy cohorts in the present MitoEAGLE BME database (average height 1.77 m). (a) Healthy reference population, HRP: thick line extrapolated by a thin line beyond the range of measurement, H = 0.411∙M°0.35 [8]. M° is the reference body mass corresponding to body height on the X-axis. The dashed line is the fit through the male and female HUNT 3 data. Vertical arrows indicate weight gain at constant body height. The green square is the Reference Man [7]. (b) The BMI with an exponent of 2 (instead of 2.86; Fig. 2b) increases with body mass in the HRP, from 18.9 to 22.9 with height increasing from 1.6 to 2.0 m. The body mass excess with respect to the HRP is defined as BME ≝ (M-M°)/M°. A balanced BME is BME°=0.0. Considering a height of 1.7 m/x (dashed horizontal lines), overweight (BMI=25) is reached at a weight gain of 20 % (BME=0.2); obesity and severe obesity (BMI=30 and 35) are reached at a weight gain of 40 % and 60 % (BME=0.4 and 0.6, respectively). (c) Mitochondrial fitness, JO2,P declines as a function of BME (MitoEAGLE BME database). JO2,P is the OXPHOS capacity of the convergent NADH- and succinate-linked pathway expressed per wet mass of muscle tissue, mw. (d) VO2max/M declines as a function of BME (MitoEAGLE BME database; a powerfunction is fitted through the open circles, shown by the full line and extrapolated to BME=0; see equation). The females of the HUNT 3 study are on the line, whereas the males tend to have a higher aerobic capacity. Dashed line (1): Aerobic capacity modelled by adding metabolically inactive body mass to the reference V°O2max/M=72.1 mL∙min-1∙kg-1. Dottel line (2): Diminishing muscle aerobic capacity according to the decline of mitochondrial fitness in Fig. 2c. Red crosses (3): A constant ‘weight-lifting’ factor is fitted to account for an increasing fraction of muscle mass as a function of BME. Modified from [10].
| Healthy reference population | Body mass excess | BFE | BME cutoffs | BMI | H | M | VO2max | mitObesity drugs |
Publications: BME and height
| Reference | |
|---|---|
| Bosy-Westphal 2009 Br J Nutr | Bosy-Westphal A, Plachta-Danielzik S, Dörhöfer RP, Müller MJ (2009) Short stature and obesity: positive association in adults but inverse association in children and adolescents. Br J Nutr 102:453-61. |
| De Onis 2007 Bull World Health Organization | de Onis M, Onyango AW, Borghi E, Siyam A, Nishida C, Siekmann J (2007) Development of a WHO growth reference for school-aged children and adolescents. Bull World Health Organization 85:660-7. |
| Gnaiger 2019 MiP2019 | OXPHOS capacity in human muscle tissue and body mass excess – the MitoEAGLE mission towards an integrative database (Version 6; 2020-01-12). |
| Hood 2019 Nutr Diabetes | Hood K, Ashcraft J, Watts K, Hong S, Choi W, Heymsfield SB, Gautam RK, Thomas D (2019) Allometric scaling of weight to height and resulting body mass index thresholds in two Asian populations. Nutr Diabetes 9:2. doi: 10.1038/s41387-018-0068-3. |
| Indian Academy of Pediatrics Growth Charts Committee 2015 Indian Pediatr | Indian Academy of Pediatrics Growth Charts Committee, Khadilkar V, Yadav S, Agrawal KK, Tamboli S, Banerjee M, Cherian A, Goyal JP, Khadilkar A, Kumaravel V, Mohan V, Narayanappa D, Ray I, Yewale V (2015) Revised IAP growth charts for height, weight and body mass index for 5- to 18-year-old Indian children. Indian Pediatr 52:47-55. |
| Zucker 1962 Committee on Biological Handbooks, Fed Amer Soc Exp Biol | Zucker TF (1962) Regression of standing and sitting weights on body weight: man. In: Growth including reproduction and morphological development. Altman PL, Dittmer DS, eds: Committee on Biological Handbooks, Fed Amer Soc Exp Biol:336-7. |
MitoPedia: BME and mitObesity
» Body mass excess and mitObesity | BME and mitObesity news | Summary |
| Term | Abbreviation | Description |
|---|---|---|
| BME cutoff points | BME cutoff | Obesity is defined as a disease associated with an excess of body fat with respect to a healthy reference condition. Cutoff points for body mass excess, BME cutoff points, define the critical values for underweight (-0.1 and -0.2), overweight (0.2), and various degrees of obesity (0.4, 0.6, 0.8, and above). BME cutoffs are calibrated by crossover-points of BME with established BMI cutoffs. |
| Body fat excess | BFE | In the healthy reference population (HRP), there is zero body fat excess, BFE, and the fraction of excess body fat in the HRP is expressed - by definition - relative to the reference body mass, M°, at any given height. Importantly, body fat excess, BFE, and body mass excess, BME, are linearly related, which is not the case for the body mass index, BMI. |
| Body mass | m [kg]; M [kg·x-1] | The body mass M is the mass (kilogram [kg]) of an individual (object) [x] and is expressed in units [kg/x]. Whereas the body weight changes as a function of gravitational force (you are weightless at zero gravity; your floating weight in water is different from your weight in air), your mass is independent of gravitational force, and it is the same in air and water. |
| Body mass excess | BME | The body mass excess, BME, is an index of obesity and as such BME is a lifestyle metric. The BME is a measure of the extent to which your actual body mass, M [kg/x], deviates from M° [kg/x], which is the reference body mass [kg] per individual [x] without excess body fat in the healthy reference population, HRP. A balanced BME is BME° = 0.0 with a band width of -0.1 towards underweight and +0.2 towards overweight. The BME is linearly related to the body fat excess. |
| Body mass index | BMI | The body mass index, BMI, is the ratio of body mass to height squared (BMI=M·H-2), recommended by the WHO as a general indicator of underweight (BMI<18.5 kg·m-2), overweight (BMI>25 kg·m-2) and obesity (BMI>30 kg·m-2). Keys et al (1972; see 2014) emphasized that 'the prime criterion must be the relative independence of the index from height'. It is exactly the dependence of the BMI on height - from children to adults, women to men, Caucasians to Asians -, which requires adjustments of BMI-cutoff points. This deficiency is resolved by the body mass excess relative to the healthy reference population. |
| Comorbidity | Comorbidities are common in obesogenic lifestyle-induced early aging. These are preventable, non-communicable diseases with strong associations to obesity. In many studies, cause and effect in the sequence of onset of comorbidities remain elusive. Chronic degenerative diseases are commonly obesity-induced. The search for the link between obesity and the etiology of diverse preventable diseases lead to the hypothesis, that mitochondrial dysfunction is the common mechanism, summarized in the term 'mitObesity'. | |
| Healthy reference population | HRP | A healthy reference population, HRP, establishes the baseline for the relation between body mass and height in healthy people of zero underweight or overweight, providing a reference for evaluation of deviations towards underweight or overweight and obesity. The WHO Child Growth Standards (WHO-CGS) on height and body mass refer to healthy girls and boys from Brazil, Ghana, India, Norway, Oman and the USA. The Committee on Biological Handbooks compiled data on height and body mass of healthy males from infancy to old age (USA), published before emergence of the fast-food and soft-drink epidemic. Four allometric phases are distinguished with distinct allometric exponents. At heights above 1.26 m/x the allometric exponent is 2.9, equal in women and men, and significantly different from the exponent of 2.0 implicated in the body mass index, BMI [kg/m2]. |
| Height of humans | h [m]; H [m·x-1] | The height of humans, h, is given in SI units in meters [m]. Humans are countable objects, and the symbol and unit of the number of objects is N [x]. The average height of N objects is, H = h/N [m/x], where h is the heights of all N objects measured on top of each other. Therefore, the height per human has the unit [m·x-1] (compare body mass [kg·x-1]). Without further identifyer, H is considered as the standing height of a human, measured without shoes, hair ornaments and heavy outer garments. |
| Length | l [m] | Length l is an SI base quantity with SI base unit meter m. Quantities derived from length are area A [m2] and volume V [m3]. Length is an extensive quantity, increasing additively with the number of objects. The term 'height' h is used for length in cases of vertical position (see height of humans). Length of height per object, LUX [m·x-1] is length per unit-entity UX, in contrast to lentgth of a system, which may contain one or many entities, such as the length of a pipeline assembled from a number NX of individual pipes. Length is a quantity linked to direct sensory, practical experience, as reflected in terms related to length: long/short (height: tall/small). Terms such as 'long/short distance' are then used by analogy in the context of the more abstract quantity time (long/short duration). |
| MitObesity drugs | Bioactive mitObesity compounds are drugs and nutraceuticals with more or less reproducible beneficial effects in the treatment of diverse preventable degenerative diseases implicated in comorbidities linked to obesity, characterized by common mechanisms of action targeting mitochondria. | |
| Obesity | Obesity is a disease resulting from excessive accumulation of body fat. In common obesity (non-syndromic obesity) excessive body fat is due to an obesogenic lifestyle with lack of physical exercise ('couch') and caloric surplus of food consumption ('potato'), causing several comorbidities which are characterized as preventable non-communicable diseases. Persistent body fat excess associated with deficits of physical activity induces a weight-lifting effect on increasing muscle mass with decreasing mitochondrial capacity. Body fat excess, therefore, correlates with body mass excess up to a critical stage of obesogenic lifestyle-induced sarcopenia, when loss of muscle mass results in further deterioration of physical performance particularly at older age. | |
| VO2max | VO2max; VO2max/M | Maximum oxygen consumption, VO2max, is and index of cardiorespiratory fitness, measured by spiroergometry on human and animal organisms capable of controlled physical exercise performance on a treadmill or cycle ergometer. VO2max is the maximum respiration of an organism, expressed as the volume of O2 at STPD consumed per unit of time per individual object [mL.min-1.x-1]. If normalized per body mass of the individual object, M [kg.x-1], mass specific maximum oxygen consumption, VO2max/M, is expressed in units [mL.min-1.kg-1]. |
Labels: MiParea: Respiration, mt-Biogenesis;mt-density, Gender, Exercise physiology;nutrition;life style, mt-Medicine
Pathology: Obesity
Organism: Human Tissue;cell: Skeletal muscle Preparation: Intact organism, Permeabilized tissue, Isolated mitochondria
Coupling state: OXPHOS
Pathway: NS
HRR: Oxygraph-2k
MitoEAGLE BME, BME, BMI, Body mass, Height, VO2max