Difference between revisions of "Gnaiger 2023 MitoFit CII"
| Line 10: | Line 10: | ||
The current narrative that the reduced coenzymes NADH and FADH<sub>2</sub> feed electrons from the tricarboxylic acid cycle into the mitochondrial electron transfer system creates ambiguities around respiratory Complex II (CII). The succinate dehydrogenase subunit SDHA of CII oxidizes succinate and reduces covalently bound FAD to FADH<sub>2</sub> in the canonical forward tricarboxylic acid cycle. However, several graphical representations of the membrane-bound electron transfer system (ETS) depict FADH<sub>2</sub> in the mitochondrial matrix to be oxidized by CII. This leads to the false conclusion that FADH<sub>2</sub> feeds electrons into the ETS through CII, including FADH<sub>2</sub> from the tricarboxylic acid cycle, the β-oxidation cycle in fatty acid oxidation, and the glycerophosphate shuttle. In reality, FAD and succinate are the ''substrates'' of SDHA at the ETS-entry into CII. The reduced flavin groups FADH<sub>2</sub> and FMNH<sub>2</sub> are ''products'' downstream within CII and CI, respectively. Further electron transfer converges at the coenzyme Q-junction. Similarly, electron transferring flavoprotein and mitochondrial glycerophosphate dehydrogenase feed electrons into the Q-junction but not through CII. The ambiguities surrounding Complex II in the literature and educational tools call for quality control, to secure scientific standards in current communications on bioenergetics and ultimately support adequate clinical applications. | The current narrative that the reduced coenzymes NADH and FADH<sub>2</sub> feed electrons from the tricarboxylic acid cycle into the mitochondrial electron transfer system creates ambiguities around respiratory Complex II (CII). The succinate dehydrogenase subunit SDHA of CII oxidizes succinate and reduces covalently bound FAD to FADH<sub>2</sub> in the canonical forward tricarboxylic acid cycle. However, several graphical representations of the membrane-bound electron transfer system (ETS) depict FADH<sub>2</sub> in the mitochondrial matrix to be oxidized by CII. This leads to the false conclusion that FADH<sub>2</sub> feeds electrons into the ETS through CII, including FADH<sub>2</sub> from the tricarboxylic acid cycle, the β-oxidation cycle in fatty acid oxidation, and the glycerophosphate shuttle. In reality, FAD and succinate are the ''substrates'' of SDHA at the ETS-entry into CII. The reduced flavin groups FADH<sub>2</sub> and FMNH<sub>2</sub> are ''products'' downstream within CII and CI, respectively. Further electron transfer converges at the coenzyme Q-junction. Similarly, electron transferring flavoprotein and mitochondrial glycerophosphate dehydrogenase feed electrons into the Q-junction but not through CII. The ambiguities surrounding Complex II in the literature and educational tools call for quality control, to secure scientific standards in current communications on bioenergetics and ultimately support adequate clinical applications. | ||
<br> | <br> | ||
|keywords=coenzyme Q junction; Complex CII; electron transfer system; fatty acid oxidation; flavin adenine dinucleotide; | |keywords=coenzyme Q junction; Complex CII; electron transfer system; fatty acid oxidation; flavin adenine dinucleotide;succinate dehydrogenase; tricarboxylic acid cycle | ||
succinate dehydrogenase; tricarboxylic acid cycle | |||
|mipnetlab=AT Innsbruck Oroboros | |mipnetlab=AT Innsbruck Oroboros | ||
}} | }} | ||
{{Labeling | |||
|additional=FAT4BRAIN, Publication:FAT4BRAIN | |||
}} | |||
[[File:N-S FADH2-FMNH2.png|right|600px]] | [[File:N-S FADH2-FMNH2.png|right|600px]] | ||
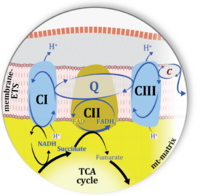
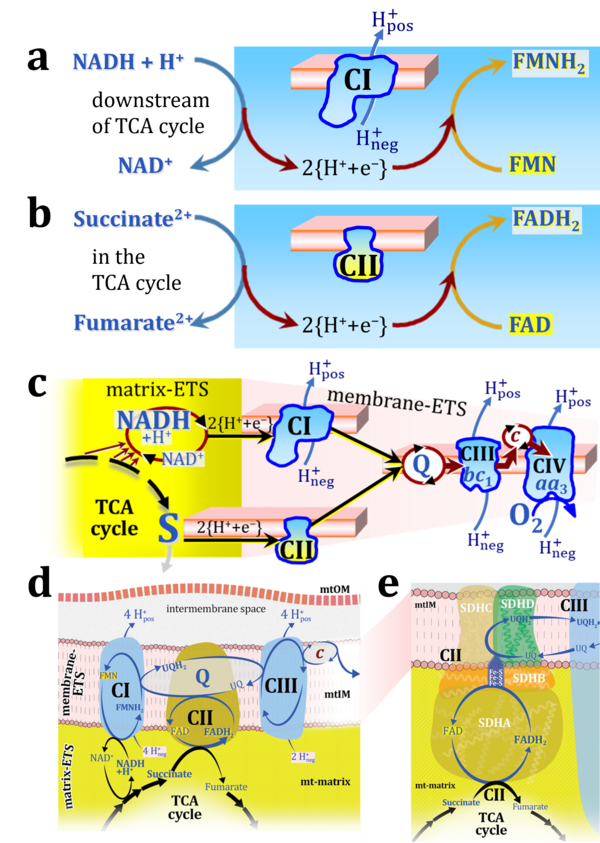
:::: '''Figure 1. Complex II bridges electron transfer from the TCA cycle to the mitochondrial inner membrane'''. Graphical representations of the electron transfer system ETS with successive emphasis on pathway architecture and concomitant loss of detail. CII is integrated in the TCA cycle (matrix-ETS) and the membrane-bound electron transfer system (membrane-ETS in the mt-inner membrane mtIM). Joint half-circular arrows indicate electron transfer 2{H<sup>+</sup>+e<sup>-</sup>}, distinguished from hydrogen ion H<sup>+</sup> transport across the mtIM. '''(a)''' In the soluble domain of CII, the flavoprotein SDHA catalyzes the oxidation succinate → fumarate+2{H<sup>+</sup>+e<sup>-</sup>} and reduction FAD+2{H<sup>+</sup>+e<sup>-</sup>} → FADH<sub>2</sub>. The iron–sulfur protein SDHB transfers electrons through Fe-S clusters to the mtIM domain where ubiquinone UQ is reduced with 2{H<sup>+</sup>+e<sup>-</sup>} to ubiquinol UQH<sub>2</sub> in SDHC and SDHD. '''(b)''' NADH and succinate are substrates of redox reactions in CI and CII, respectively, with FMNH<sub>2</sub> and FADH<sub>2</sub> as the corresponding products. Succinate and fumarate indicate the chemical entities irrespective of ionization, whereas the charges are shown in NADH, NAD<sup>+</sup>, and H<sup>+</sup>. '''(c)''' Electron flow catalyzed by dehydrognases localized in the mitochondrial (mt) matrix converges at the N-junction, reducing NAD<sup>+</sup> to NADH. Electron flow from NADH and succinate S to molecular oxygen, 2{H<sup>+</sup>+e<sup>-</sup>}+0.5 O<sub>2</sub> ⇢ H<sub>2</sub>O, converges through CI and CII at the Q-junction. CIII passes electrons to cytochrome ''c'' and in CIV to O<sub>2</sub>. | :::: '''Figure 1. Complex II bridges electron transfer from the TCA cycle to the mitochondrial inner membrane'''. Graphical representations of the electron transfer system ETS with successive emphasis on pathway architecture and concomitant loss of detail. CII is integrated in the TCA cycle (matrix-ETS) and the membrane-bound electron transfer system (membrane-ETS in the mt-inner membrane mtIM). Joint half-circular arrows indicate electron transfer 2{H<sup>+</sup>+e<sup>-</sup>}, distinguished from hydrogen ion H<sup>+</sup> transport across the mtIM. '''(a)''' In the soluble domain of CII, the flavoprotein SDHA catalyzes the oxidation succinate → fumarate+2{H<sup>+</sup>+e<sup>-</sup>} and reduction FAD+2{H<sup>+</sup>+e<sup>-</sup>} → FADH<sub>2</sub>. The iron–sulfur protein SDHB transfers electrons through Fe-S clusters to the mtIM domain where ubiquinone UQ is reduced with 2{H<sup>+</sup>+e<sup>-</sup>} to ubiquinol UQH<sub>2</sub> in SDHC and SDHD. '''(b)''' NADH and succinate are substrates of redox reactions in CI and CII, respectively, with FMNH<sub>2</sub> and FADH<sub>2</sub> as the corresponding products. Succinate and fumarate indicate the chemical entities irrespective of ionization, whereas the charges are shown in NADH, NAD<sup>+</sup>, and H<sup>+</sup>. '''(c)''' Electron flow catalyzed by dehydrognases localized in the mitochondrial (mt) matrix converges at the N-junction, reducing NAD<sup>+</sup> to NADH. Electron flow from NADH and succinate S to molecular oxygen, 2{H<sup>+</sup>+e<sup>-</sup>}+0.5 O<sub>2</sub> ⇢ H<sub>2</sub>O, converges through CI and CII at the Q-junction. CIII passes electrons to cytochrome ''c'' and in CIV to O<sub>2</sub>. | ||
| Line 543: | Line 544: | ||
:::::: [[File:Expii-Gabi Slizewska CORRECTION.png|400px]] | :::::: [[File:Expii-Gabi Slizewska CORRECTION.png|400px]] | ||
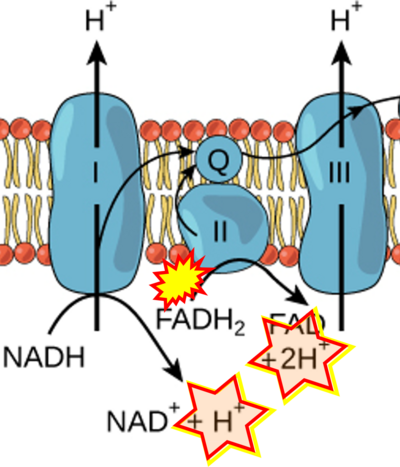
:::: '''Website 9''': [https://www.expii.com/t/electron-transport-chain-summary-diagrams-10139 expii] - Image source: By Gabi Slizewska | :::: '''Website 9''': [https://www.expii.com/t/electron-transport-chain-summary-diagrams-10139 expii] - Image source: By Gabi Slizewska | ||
Revision as of 12:06, 17 April 2023
| Gnaiger E (2023) Complex II ambiguities ― FADH2 in the electron transfer system. MitoFit Preprints 2023.3.v2. https://doi.org/10.26124/mitofit:2023-0003.v2 |
» MitoFit Preprints 2023.3.v2.
Complex II ambiguities ― FADH2 in the electron transfer system
Gnaiger Erich (2023) MitoFit Prep
Abstract:
- Version 2 (v2) 2023-04-04 10.26124/mitofit:2023-0003.v2
- Version 1 (v1) 2023-03-247 10.26124/mitofit:2023-0003 - »Link to all versions«
The current narrative that the reduced coenzymes NADH and FADH2 feed electrons from the tricarboxylic acid cycle into the mitochondrial electron transfer system creates ambiguities around respiratory Complex II (CII). The succinate dehydrogenase subunit SDHA of CII oxidizes succinate and reduces covalently bound FAD to FADH2 in the canonical forward tricarboxylic acid cycle. However, several graphical representations of the membrane-bound electron transfer system (ETS) depict FADH2 in the mitochondrial matrix to be oxidized by CII. This leads to the false conclusion that FADH2 feeds electrons into the ETS through CII, including FADH2 from the tricarboxylic acid cycle, the β-oxidation cycle in fatty acid oxidation, and the glycerophosphate shuttle. In reality, FAD and succinate are the substrates of SDHA at the ETS-entry into CII. The reduced flavin groups FADH2 and FMNH2 are products downstream within CII and CI, respectively. Further electron transfer converges at the coenzyme Q-junction. Similarly, electron transferring flavoprotein and mitochondrial glycerophosphate dehydrogenase feed electrons into the Q-junction but not through CII. The ambiguities surrounding Complex II in the literature and educational tools call for quality control, to secure scientific standards in current communications on bioenergetics and ultimately support adequate clinical applications.
• Keywords: coenzyme Q junction; Complex CII; electron transfer system; fatty acid oxidation; flavin adenine dinucleotide;succinate dehydrogenase; tricarboxylic acid cycle
• O2k-Network Lab: AT Innsbruck Oroboros
Labels:
FAT4BRAIN, Publication:FAT4BRAIN
- Figure 1. Complex II bridges electron transfer from the TCA cycle to the mitochondrial inner membrane. Graphical representations of the electron transfer system ETS with successive emphasis on pathway architecture and concomitant loss of detail. CII is integrated in the TCA cycle (matrix-ETS) and the membrane-bound electron transfer system (membrane-ETS in the mt-inner membrane mtIM). Joint half-circular arrows indicate electron transfer 2{H++e-}, distinguished from hydrogen ion H+ transport across the mtIM. (a) In the soluble domain of CII, the flavoprotein SDHA catalyzes the oxidation succinate → fumarate+2{H++e-} and reduction FAD+2{H++e-} → FADH2. The iron–sulfur protein SDHB transfers electrons through Fe-S clusters to the mtIM domain where ubiquinone UQ is reduced with 2{H++e-} to ubiquinol UQH2 in SDHC and SDHD. (b) NADH and succinate are substrates of redox reactions in CI and CII, respectively, with FMNH2 and FADH2 as the corresponding products. Succinate and fumarate indicate the chemical entities irrespective of ionization, whereas the charges are shown in NADH, NAD+, and H+. (c) Electron flow catalyzed by dehydrognases localized in the mitochondrial (mt) matrix converges at the N-junction, reducing NAD+ to NADH. Electron flow from NADH and succinate S to molecular oxygen, 2{H++e-}+0.5 O2 ⇢ H2O, converges through CI and CII at the Q-junction. CIII passes electrons to cytochrome c and in CIV to O2.
ORCID: ![]() Gnaiger Erich, Oroboros Instruments, Innsbruck, Austria
Gnaiger Erich, Oroboros Instruments, Innsbruck, Austria
- Acknowledgements: I thank Luiza H. Cardoso and Sabine Schmitt for stimulating discussions, and Paolo Cocco for expert help on the graphical abstract and Figures 1b and c. Contribution to the European Union’s Horizon 2020 research and innovation program Grant 857394 (FAT4BRAIN).
Updates to Supplement Figure S1
Last update 2023-04-15
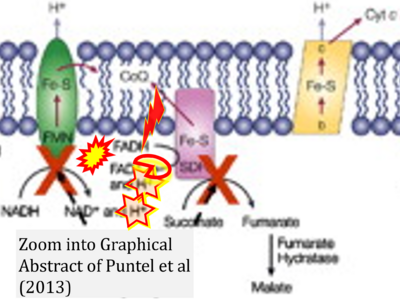
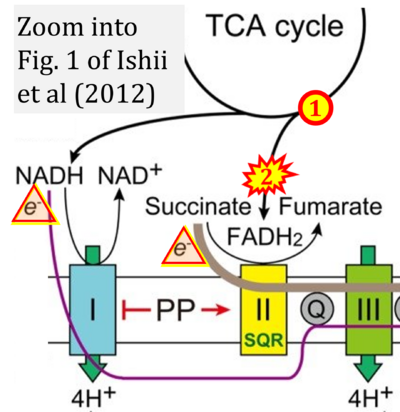
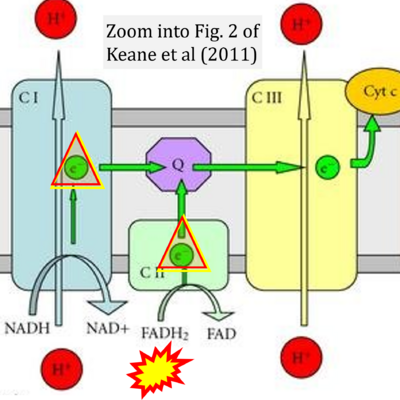
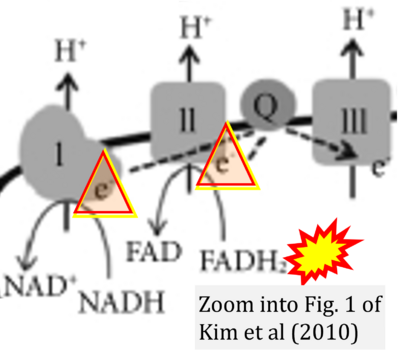
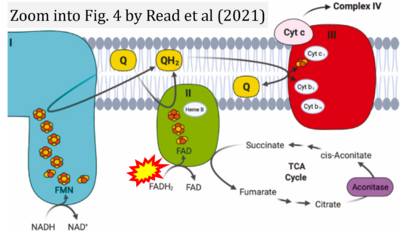
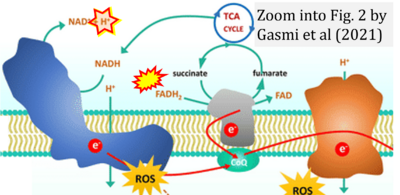
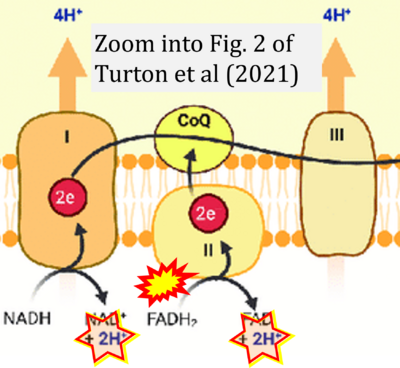
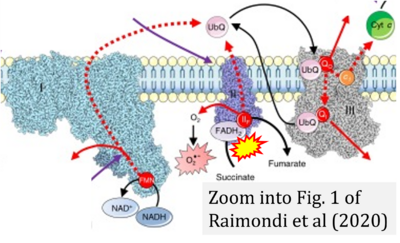
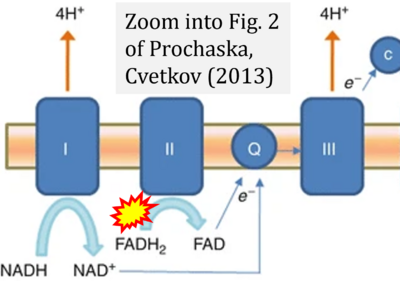
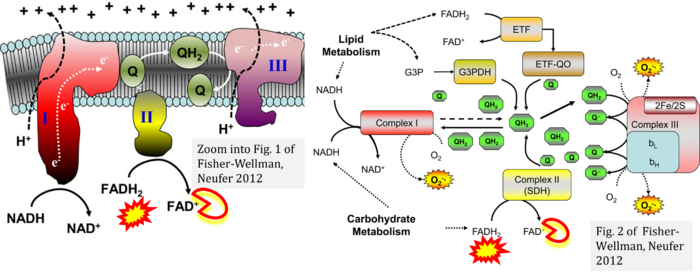
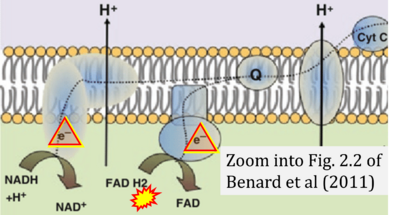
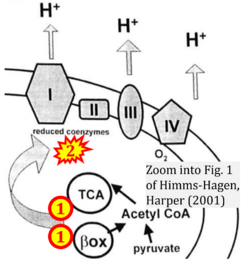
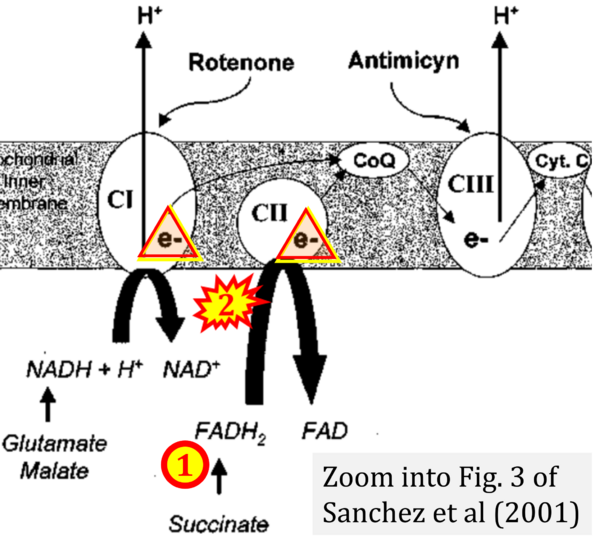
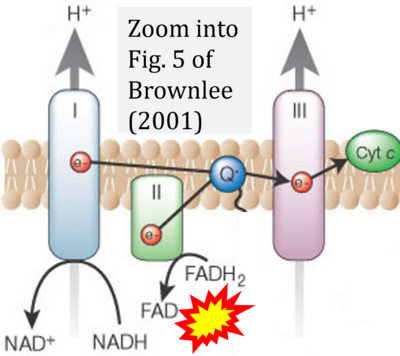
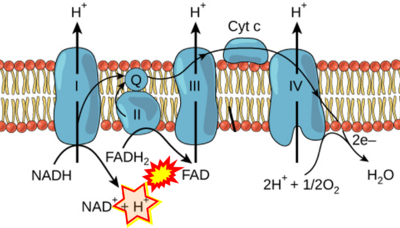
- Figure S1. Complex II ambiguities in graphical representations on FADH2 as a substrate of Complex II in the canonical forward electron transfer. Chronological sequence of publications from 2001 to 2023.
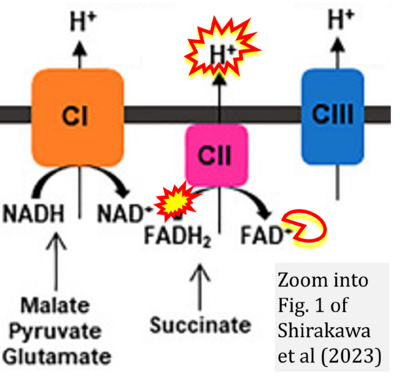
- 1 Shirakawa R, Nakajima T, Yoshimura A, Kawahara Y, Orito C, Yamane M, Handa H, Takada S, Furihata T, Fukushima A, Ishimori N, Nakagawa M, Yokota I, Sabe H, Hashino S, Kinugawa S, Yokota T (2023) Enhanced mitochondrial oxidative metabolism in peripheral blood mononuclear cells is associated with fatty liver in obese young adults. Sci Rep 13:5203. - »Bioblast link«
- While CI functions as a proton pump, CII does not. Depicting CII as a proton pump would be analogous to falsely portraying FADH2 as the substrate of CII, as if it were a copy of CI, which functions as a proton pump with NADH as its substrate.
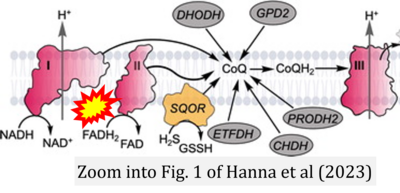
- 2 Hanna D, Kumar R, Banerjee R (2023) A metabolic paradigm for hydrogen sulfide signaling via electron transport chain plasticity. Antioxid Redox Signal 38:57-67. - »Bioblast link«
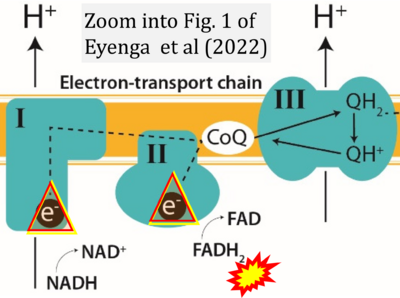
- 3 Eyenga P, Rey B, Eyenga L, Sheu SS (2022) Regulation of oxidative phosphorylation of liver mitochondria in sepsis. Cells 11:1598. - »Bioblast link«
- 4 Tseng W-W, Wei A-C (2022) Kinetic mathematical modeling of oxidative phosphorylation in cardiomyocyte mitochondria. Cells 11:4020. - »Bioblast link«
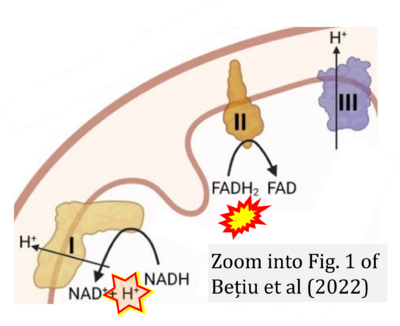
- 5 Bețiu AM, Noveanu L, Hâncu IM, Lascu A, Petrescu L, Maack C, Elmér E, Muntean DM (2022) Mitochondrial effects of common cardiovascular medications: the good, the bad and the mixed. Int J Mol Sci 23:13653. - »Bioblast link«
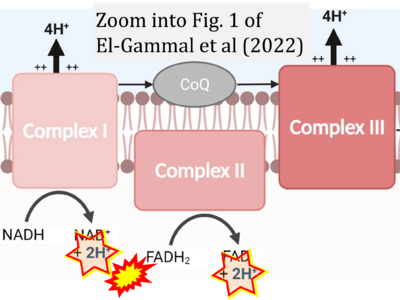
- 6 El-Gammal Z, Nasr MA, Elmehrath AO, Salah RA, Saad SM, El-Badri N (2022) Regulation of mitochondrial temperature in health and disease. Pflugers Arch 474:1043-51. - »Bioblast link«
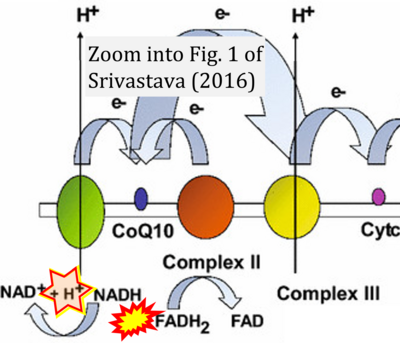
- 7 Peng M, Huang Y, Zhang L, Zhao X, Hou Y (2022) Targeting mitochondrial oxidative phosphorylation eradicates acute myeloid leukemic stem cells. Front Oncol 12:899502. - »Bioblast link«
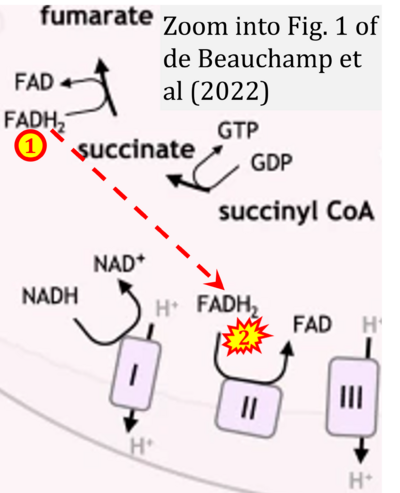
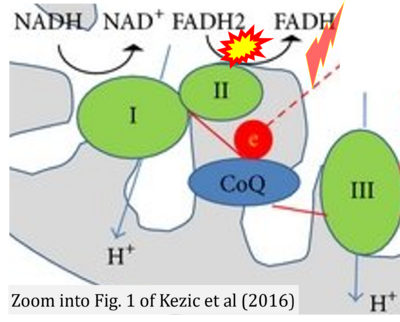
- 8 de Beauchamp L, Himonas E, Helgason GV (2022) Mitochondrial metabolism as a potential therapeutic target in myeloid leukaemia. Leukemia 36:1-12. - »Bioblast link«
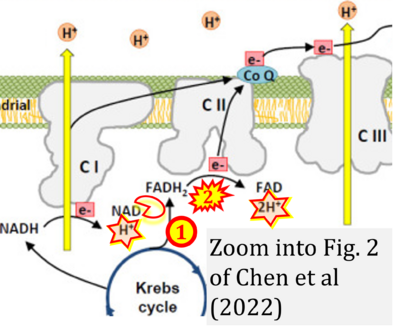
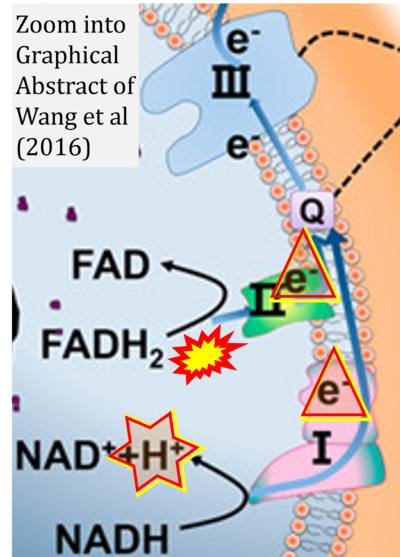
- 9 Chen TH, Koh KY, Lin KM, Chou CK (2022) Mitochondrial dysfunction as an underlying cause of skeletal muscle disorders. Int J Mol Sci 23:12926. - »Bioblast link«
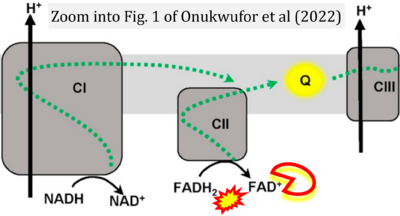
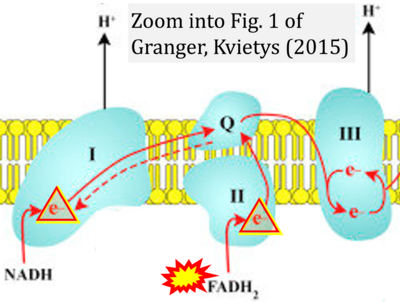
- 10 Onukwufor JO, Dirksen RT, Wojtovich AP (2022) Iron dysregulation in mitochondrial dysfunction and Alzheimer's disease. Antioxidants (Basel) 11:692. - »Bioblast link«
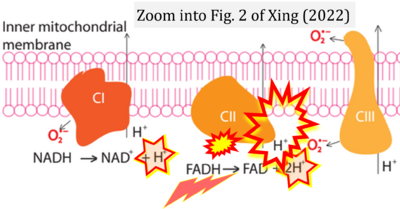
- 11 Xing Yunxie (2022) Is genome instability a significant cause of aging? A review. Atlantis Press. - »Bioblast link«
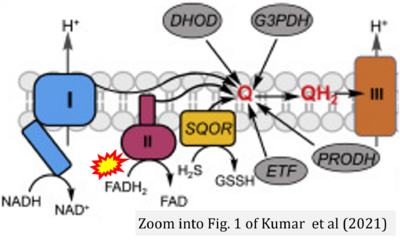
- 12 Kumar R, Landry AP, Guha A, Vitvitsky V, Lee HJ, Seike K, Reddy P, Lyssiotis CA, Banerjee R (2021) A redox cycle with complex II prioritizes sulfide quinone oxidoreductase dependent H2S oxidation. J Biol Chem 298:101435. - »Bioblast link«
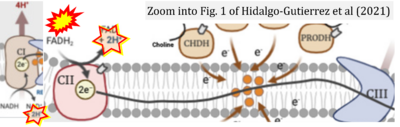
- 13 Hidalgo-Gutiérrez A, González-García P, Díaz-Casado ME, Barriocanal-Casado E, López-Herrador S, Quinzii CM, López LC (2021) Metabolic targets of coenzyme Q10 in mitochondria. Antioxidants (Basel) 10:520. - »Bioblast link«
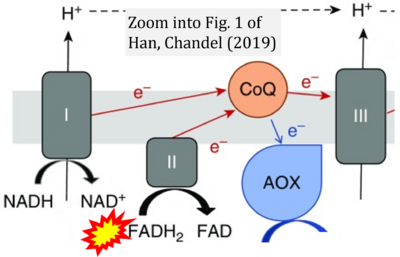
- 14 Han S, Chandel NS (2021) Lessons from cancer metabolism for pulmonary arterial hypertension and fibrosis. Am J Respir Cell Mol Biol 65:134-45. - »Bioblast link«
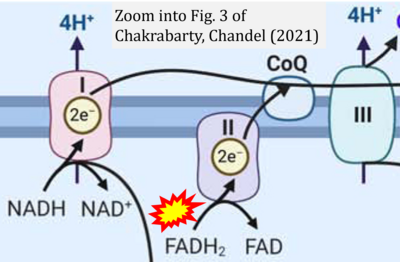
- 15 Chakrabarty RP, Chandel NS (2021) Mitochondria as signaling organelles control mammalian stem cell fate. Cell Stem Cell 28:394-408. - »Bioblast link«
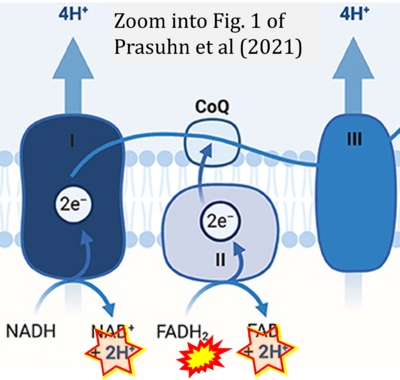
- 16 Prasuhn J, Davis RL, Kumar KR (2021) Targeting mitochondrial impairment in Parkinson's disease: challenges and opportunities. Front Cell Dev Biol 8:615461. - »Bioblast link«
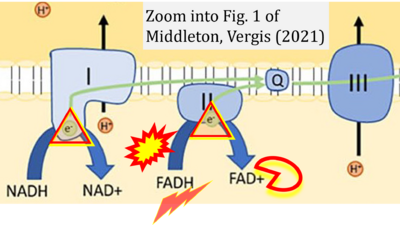
- 17 Middleton P, Vergis N (2021) Mitochondrial dysfunction and liver disease: role, relevance, and potential for therapeutic modulation. Therap Adv Gastroenterol 14:17562848211031394. - »Bioblast link«
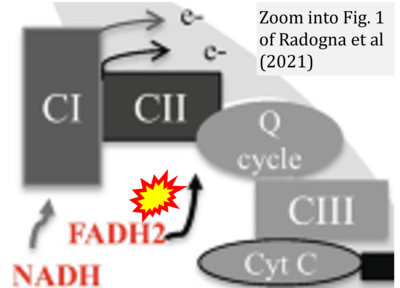
- 18 Radogna F, Gerard D, Dicato M, Diederich M (2021) Assessment of mitochondrial cell metabolism by respiratory chain electron flow assays. Methods Mol Biol 2276:129-141. - »Bioblast link«
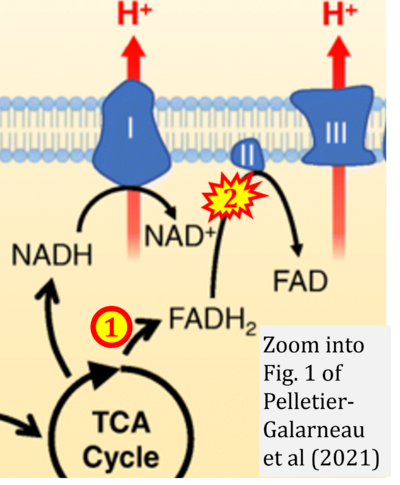
- 19 Pelletier-Galarneau M, Detmer FJ, Petibon Y, Normandin M, Ma C, Alpert NM, El Fakhri G (2021) Quantification of myocardial mitochondrial membrane potential using PET. Curr Cardiol Rep 23:70. - »Bioblast link«
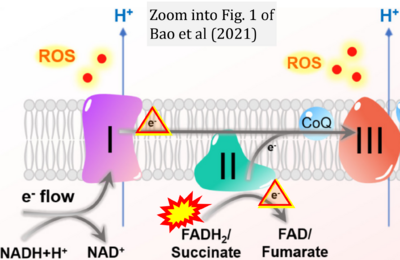
- 20 Bao MH, Wong CC (2021) Hypoxia, metabolic reprogramming, and drug resistance in liver cancer. Cells 10:1715. - »Bioblast link«
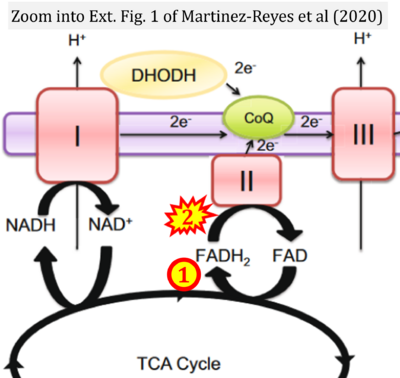
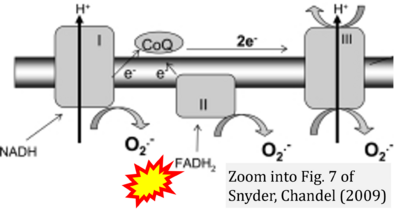
- 21 Martínez-Reyes I, Cardona LR, Kong H, Vasan K, McElroy GS, Werner M, Kihshen H, Reczek CR, Weinberg SE, Gao P, Steinert EM, Piseaux R, Budinger GRS, Chandel NS (2020) Mitochondrial ubiquinol oxidation is necessary for tumour growth. Nature 585:288-92. - »Bioblast link«
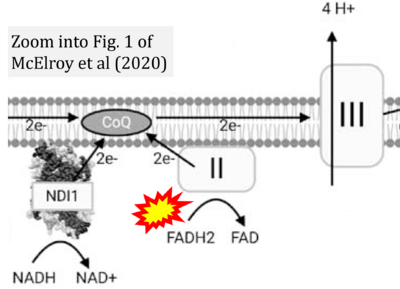
- 22 McElroy GS, Reczek CR, Reyfman PA, Mithal DS, Horbinski CM, Chandel NS (2020) NAD+ regeneration rescues lifespan, but not ataxia, in a mouse model of brain mitochondrial Complex I dysfunction. Cell Metab 32:301-8.e6. - »Bioblast link«
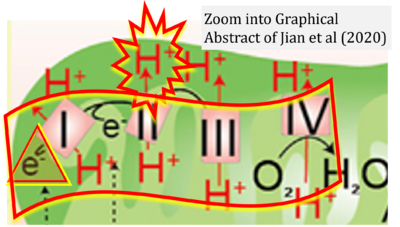
- 23 Jian C, Fu J, Cheng X, Shen LJ, Ji YX, Wang X, Pan S, Tian H, Tian S, Liao R, Song K, Wang HP, Zhang X, Wang Y, Huang Z, She ZG, Zhang XJ, Zhu L, Li H (2020) Low-dose sorafenib acts as a mitochondrial uncoupler and ameliorates nonalcoholic steatohepatitis. Cell Metab 31:892-908. - »Bioblast link«
- While CI functions as a proton pump, CII does not. Depicting CII as a proton pump would be analogous to falsely portraying FADH2 as the substrate of CII, as if it were a copy of CI, which functions as a proton pump with NADH as its substrate.
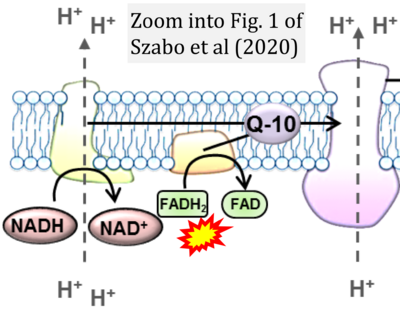
- 24 Szabo L, Eckert A, Grimm A (2020) Insights into disease-associated tau impact on mitochondria. Int J Mol Sci 21:6344. - »Bioblast link«
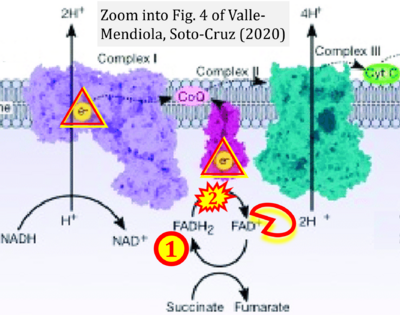
- 25 Valle-Mendiola A, Soto-Cruz I (2020) Energy metabolism in cancer: The roles of STAT3 and STAT5 in the regulation of metabolism-related genes. Cancers (Basel) 12:124. - »Bioblast link«
- 26 Tabassum N, Kheya IS, Ibn Asaduzzaman SA, Maniha SM, Fayz AH, Zakaria A, Fayz AH, Zakaria A, Noor R (2020) A review on the possible leakage of electrons through the electron transport chain within mitochondria. J Biomed Res Environ Sci 1:105-13. - »Bioblast link«
- 27 Yang L, Youngblood H, Wu C, Zhang Q (2020) Mitochondria as a target for neuroprotection: role of methylene blue and photobiomodulation. Transl Neurodegener 9:19. - »Bioblast link«
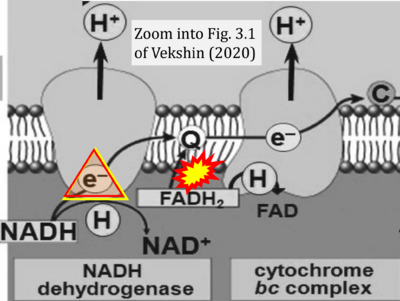
- 28 Vekshin N (2020) Biophysics of mitochondria. Springer Cham: 197 pp. - »Bioblast link«
- 29 Han S, Chandel NS (2019) There is no smoke without mitochondria. Am J Respir Cell Mol Biol 60:489-91. - »Bioblast link«
- 30 Grandoch M, Flögel U, Virtue S, Maier JK, Jelenik T, Kohlmorgen C, Feldmann K, Ostendorf Y, Castañeda TR, Zhou Z, Yamaguchi Y, Nascimento EBM, Sunkari VG, Goy C, Kinzig M, Sörgel F, Bollyky PL, Schrauwen P, Al-Hasani H, Roden M, Keipert S, Vidal-Puig A, Jastroch M5, Haendeler J, Fischer JW (2019) 4-Methylumbelliferone improves the thermogenic capacity of brown adipose tissue. Nat Metab 1:546-59. - »Bioblast link«
- NADH is shown as the product of the reaction catalyzed by CI in respiration. This error is rare in the literature, but comparable to the error frequenty encountered when FADH2 is shown as the substrate of CII.
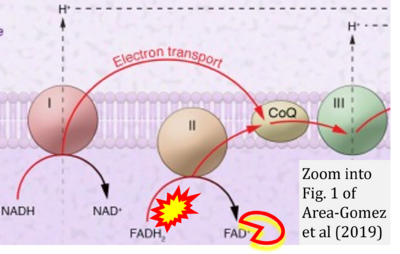
- 31 Area-Gomez E, Guardia-Laguarta C, Schon EA, Przedborski S (2019) Mitochondria, OxPhos, and neurodegeneration: cells are not just running out of gas. J Clin Invest 129:34-45. - »Bioblast link«
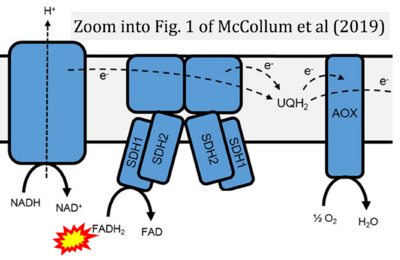
- 32 McCollum C, Geißelsöder S, Engelsdorf T, Voitsik AM, Voll LM (2019) Deficiencies in the mitochondrial electron transport chain affect redox poise and resistance toward Colletotrichum higginsianum. Front Plant Sci 10:1262. - »Bioblast link«
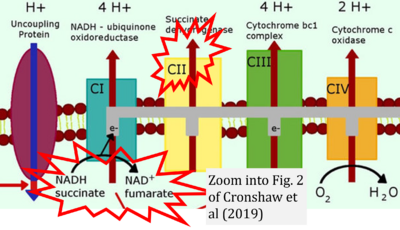
- 33 Cronshaw M, Parker S, Arany P (2019) Feeling the heat: evolutionary and microbial basis for the analgesic mechanisms of photobiomodulation therapy. Photobiomodul Photomed Laser Surg 37:517-26. - »Bioblast link«
- 34 Payen VL, Zampieri LX, Porporato PE, Sonveaux P (2019) Pro- and antitumor effects of mitochondrial reactive oxygen species. Cancer Metastasis Rev 38:189-203. - »Bioblast link«
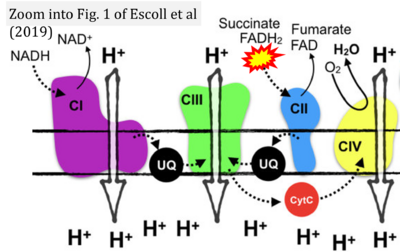
- 35 Escoll P, Platon L, Buchrieser C (2023) Roles of mitochondrial respiratory Complexes during infection. Immunometabolism 1:e190011. - »Bioblast link«
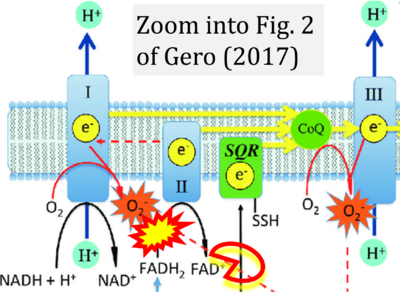
- 36 Gero D (2023) Hyperglycemia-induced endothelial dysfunction. IntechOpen Chapter 8. - »Bioblast link«
- 37 Rodick TC, Seibels DR, Babu JR, Huggins KW, Ren G, Mathews ST (2018) Potential role of coenzyme Q10 in health and disease conditions. Nutrition and Dietary Supplements 10:1-11. - »Bioblast link«
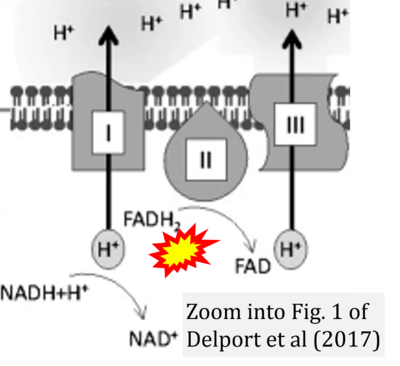
- 38 Delport A, Harvey BH, Petzer A, Petzer JP (2017) Methylene blue and its analogues as antidepressant compounds. Metab Brain Dis 32:1357-82. - »Bioblast link«
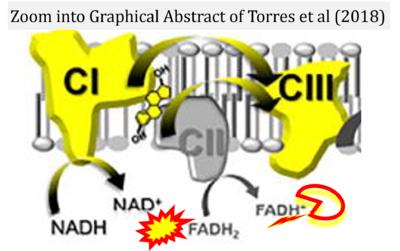
- 39 Torres MJ, Kew KA, Ryan TE, Pennington ER, Lin CT, Buddo KA, Fix AM, Smith CA, Gilliam LA, Karvinen S, Lowe DA, Spangenburg EE, Zeczycki TN, Shaikh SR, Neufer PD (2017) 17β-estradiol directly lowers mitochondrial membrane microviscosity and improves bioenergetic function in skeletal muscle. Cell Metab 27:167-79. - »Bioblast link«
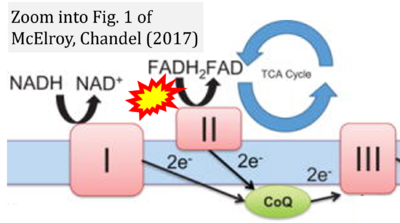
- 40 McElroy GS, Chandel NS (2017) Mitochondria control acute and chronic responses to hypoxia. Exp Cell Res 356:217-22. - »Bioblast link«
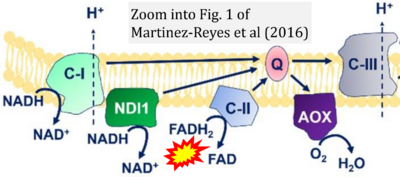
- 41 Martínez-Reyes I, Diebold LP, Kong H, Schieber M, Huang H, Hensley CT, Mehta MM, Wang T, Santos JH, Woychik R, Dufour E, Spelbrink JN, Weinberg SE, Zhao Y, DeBerardinis RJ, Chandel NS (2016) TCA cycle and mitochondrial membrane potential are necessary for diverse biological functions. Mol Cell 61:199-209. - »Bioblast link«
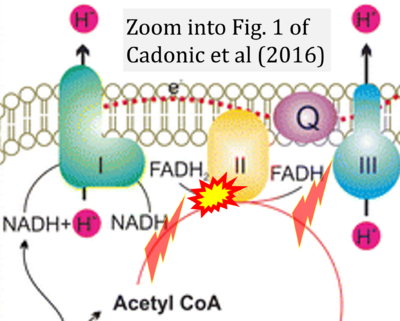
- 42 Cadonic C, Sabbir MG, Albensi BC (2016) Mechanisms of mitochondrial dysfunction in Alzheimer's disease. Mol Neurobiol 53:6078-90. - »Bioblast link«
- 43 Srivastava S (2016) Emerging therapeutic roles for NAD(+) metabolism in mitochondrial and age-related disorders. Clin Transl Med 5:25. - »Bioblast link«
- 44 Kezic A, Spasojevic I, Lezaic V, Bajcetic M (2016) Mitochondria-targeted antioxidants: future perspectives in kidney ischemia reperfusion injury. Oxid Med Cell Longev 2016:2950503. - »Bioblast link«
- 45 Wang G, Feng H, Gao A, Hao Q, Jin W, Peng X, Li W, Wu G, Chu PK (2016) Extracellular electron transfer from aerobic bacteria to Au-loaded TiO2 semiconductor without light: a new bacteria-killing mechanism other than localized surface plasmon resonance or microbial fuel cells. ACS Appl Mater Interfaces 8:24509-16. - »Bioblast link«
- 46 Granger DN, Kvietys PR (2015) Reperfusion injury and reactive oxygen species: The evolution of a concept. Redox Biol 6:524-551. - »Bioblast link«
- 47 Sullivan LB, Chandel NS (2014) Mitochondrial metabolism in TCA cycle mutant cancer cells. Cell Cycle 13:347-8. - »Bioblast link«
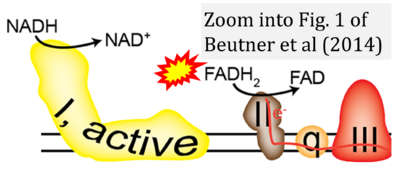
- 48 Beutner G, Eliseev RA, Porter GA Jr (2014) Initiation of electron transport chain activity in the embryonic heart coincides with the activation of mitochondrial complex 1 and the formation of supercomplexes. PLoS One 9:e113330. - »Bioblast link«
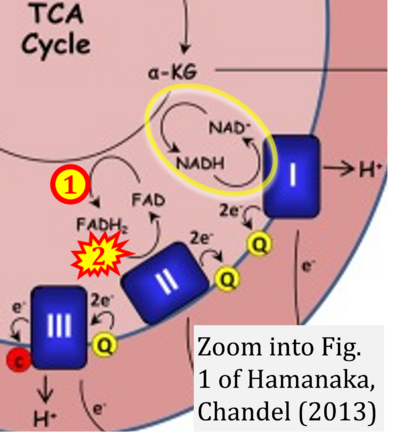
- 49 Hamanaka RB, Chandel NS (2013) Mitochondrial metabolism as a regulator of keratinocyte differentiation. Cell Logist 3:e25456. - »Bioblast link«
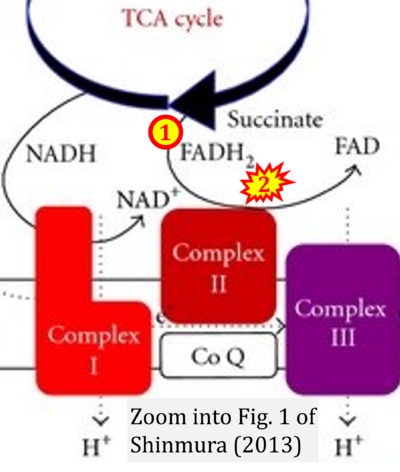
- 50 Shinmura K (2013) Effects of caloric restriction on cardiac oxidative stress and mitochondrial bioenergetics: potential role of cardiac sirtuins. Oxid Med Cell Longev 2013:528935. - »Bioblast link«
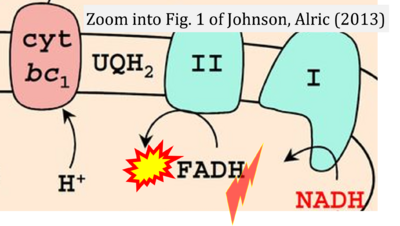
- 51 Johnson X, Alric J (2013) Central carbon metabolism and electron transport in Chlamydomonas reinhardtii: metabolic constraints for carbon partitioning between oil and starch. Eukaryot Cell 12:776-93. - »Bioblast link«
- 52 Li X, Fang P, Mai J, Choi ET, Wang H, Yang XF (2013) Targeting mitochondrial reactive oxygen species as novel therapy for inflammatory diseases and cancers. J Hematol Oncol 6:19. - »Bioblast link«
- 53 Puntel RL, Roos DH, Seeger RL, Rocha JB (2013) Mitochondrial electron transfer chain complexes inhibition by different organochalcogens. Toxicol In Vitro 27:59-70. - »Bioblast link«
- 54 Ishii I, Harada Y, Kasahara T (2012) Reprofiling a classical anthelmintic, pyrvinium pamoate, as an anti-cancer drug targeting mitochondrial respiration. Front Oncol 2:137. - »Bioblast link«
- 55 Keane PC, Kurzawa M, Blain PG, Morris CM (2011) Mitochondrial dysfunction in Parkinson's disease. Parkinsons Dis 2011:716871. - »Bioblast link«
- 56 Kim EH, Koh EH, Park JY, Lee KU (2010) Adenine nucleotide translocator as a regulator of mitochondrial function: implication in the pathogenesis of metabolic syndrome. Korean Diabetes J 34:146-53. - »Bioblast link«
- 57 Hamanaka RB, Chandel NS (2013) Snyder CM, Chandel NS (2009) Mitochondrial regulation of cell survival and death during low-oxygen conditions. Antioxid Redox Signal 11:2673-83. - »Bioblast link«
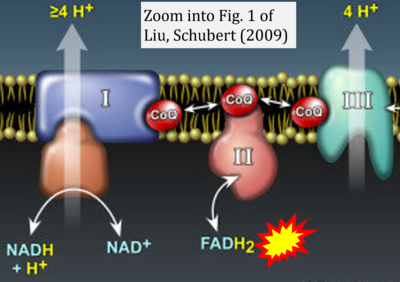
- 58 Liu Y, Schubert DR (2009) The specificity of neuroprotection by antioxidants. J Biomed Sci 16:98. - »Bioblast link«
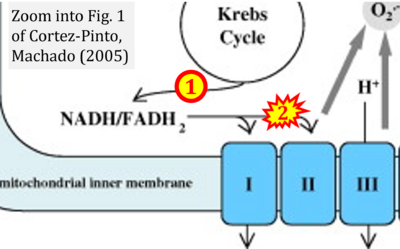
- 59 Cortez-Pinto H, Machado MV (2009) Uncoupling proteins and non-alcoholic fatty liver disease. J Hepatol 50:857-60. - »Bioblast link«
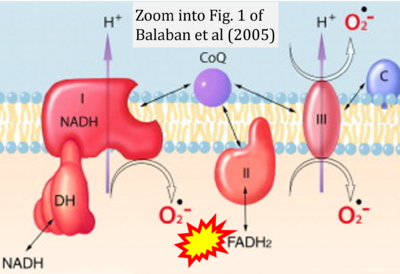
- 60 Balaban RS, Nemoto S, Finkel T (2005) Mitochondria, oxidants, and aging. Cell 120:483-95. - »Bioblast link«
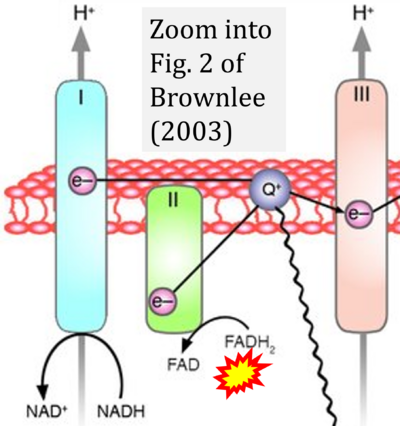
- 61 Brownlee M (2003) A radical explanation for glucose-induced beta cell dysfunction. J Clin Invest 112:1788-90. - »Bioblast link«
Supplement Figure S1 (v2)
- Figure S1. Complex II ambiguities in graphical representations on FADH2 as a substrate of Complex II in the canonical forward electron transfer. Chronological sequence of publications from 2001 to 2023.
- a Arnold PK, Finley LWS (2023) Regulation and function of the mammalian tricarboxylic acid cycle. J Biol Chem 299:102838. - »Bioblast link«
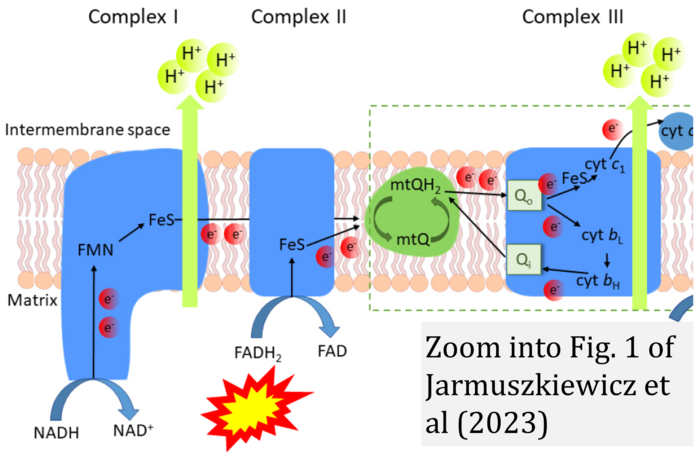
- b Jarmuszkiewicz W, Dominiak K, Budzinska A, Wojcicki K, Galganski L (2023) Mitochondrial coenzyme Q redox homeostasis and reactive oxygen species production. Front Biosci (Landmark Ed) 28:61. - »Bioblast link«
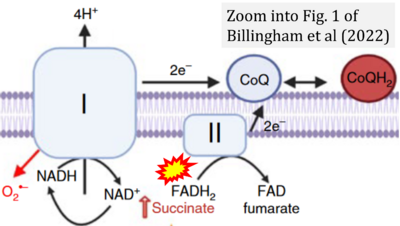
- c Billingham LK, Stoolman JS, Vasan K, Rodriguez AE, Poor TA, Szibor M, Jacobs HT, Reczek CR, Rashidi A, Zhang P, Miska J, Chandel NS (2022) Mitochondrial electron transport chain is necessary for NLRP3 inflammasome activation. Nat Immunol 23:692-704. - »Bioblast link«
- d Chen CL, Zhang L, Jin Z, Kasumov T, Chen YR (2022) Mitochondrial redox regulation and myocardial ischemia-reperfusion injury. Am J Physiol Cell Physiol 322:C12-23. - »Bioblast link«
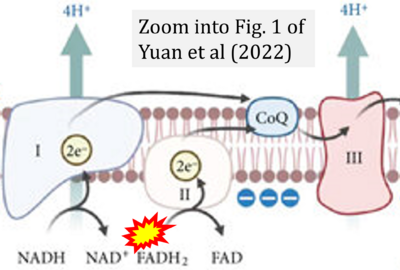
- e Yuan Q, Zeng ZL, Yang S, Li A, Zu X, Liu J (2022) Mitochondrial stress in metabolic inflammation: modest benefits and full losses. Oxid Med Cell Longev 2022:8803404. - »Bioblast link«
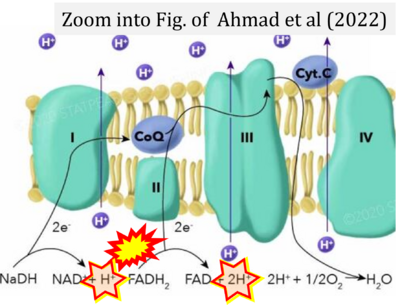
- f Ahmad M, Wolberg A, Kahwaji CI (2022) Biochemistry, electron transport chain. StatPearls Publishing StatPearls [Internet]. Treasure Island (FL) - »Bioblast link«
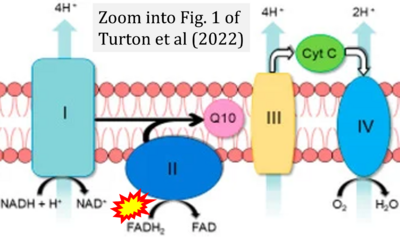
- g Turton N, Cufflin N, Dewsbury M, Fitzpatrick O, Islam R, Watler LL, McPartland C, Whitelaw S, Connor C, Morris C, Fang J, Gartland O, Holt L, Hargreaves IP (2022) The biochemical assessment of mitochondrial respiratory chain disorders. Int J Mol Sci 23:7487. - »Bioblast link«
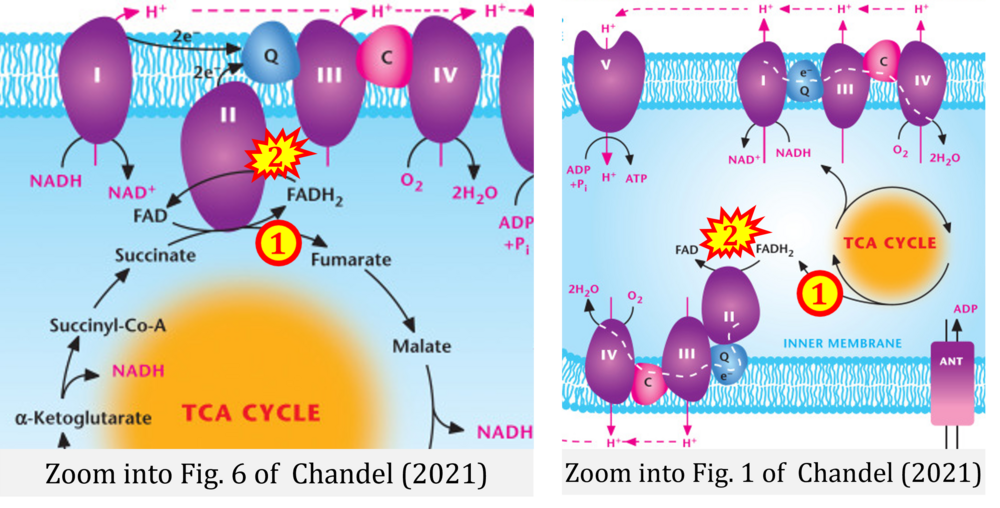
- h Chandel NS (2021) Mitochondria. Cold Spring Harb Perspect Biol 13:a040543. - »Bioblast link«
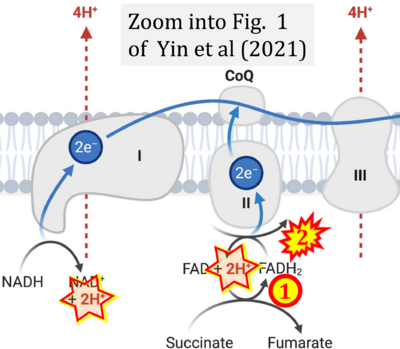
- i Yin M, O'Neill LAJ (2021) The role of the electron transport chain in immunity. FASEB J 35:e21974. - »Bioblast link«
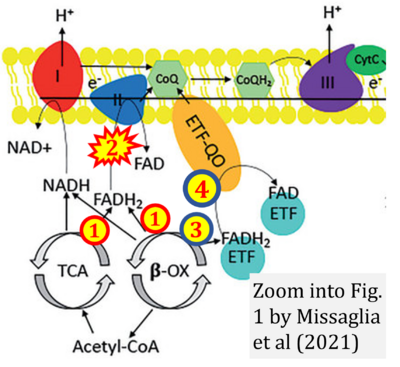
- j Missaglia S, Tavian D, Angelini C (2021) ETF dehydrogenase advances in molecular genetics and impact on treatment. Crit Rev Biochem Mol Biol 56:360-72. - »Bioblast link«
- k Read AD, Bentley RE, Archer SL, Dunham-Snary KJ (2021) Mitochondrial iron-sulfur clusters: Structure, function, and an emerging role in vascular biology. Redox Biol 47:102164. - »Bioblast link«
- l Gasmi A, Peana M, Arshad M, Butnariu M, Menzel A, Bjørklund G (2021) Krebs cycle: activators, inhibitors and their roles in the modulation of carcinogenesis. Arch Toxicol 95:1161-78. - »Bioblast link«
- m Turton N, Bowers N, Khajeh S, Hargreaves IP, Heaton RA (2021) Coenzyme Q10 and the exclusive club of diseases that show a limited response to treatment. Expert Opinion Orphan Drugs 9:151-60. - »Bioblast link«
- n Martínez-Reyes I, Chandel NS (2020) Mitochondrial TCA cycle metabolites control physiology and disease. Nat Commun 11:102. - »Bioblast link«
- o Raimondi V, Ciccarese F, Ciminale V (2020) Oncogenic pathways and the electron transport chain: a dangeROS liaison. Br J Cancer 122:168-81. - »Bioblast link«
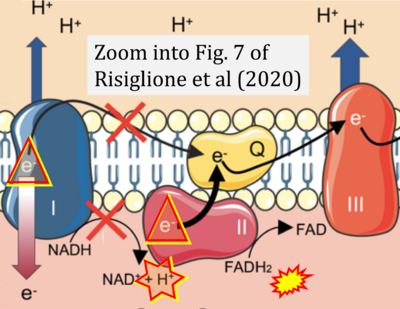
- p Risiglione P, Leggio L, Cubisino SAM, Reina S, Paternò G, Marchetti B, Magrì A, Iraci N, Messina A (2020) High-resolution respirometry reveals MPP+ mitochondrial toxicity mechanism in a cellular model of parkinson's disease. Int J Mol Sci 21:E7809. - »Bioblast link«
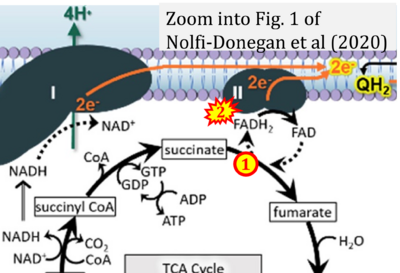
- q Nolfi-Donegan D, Braganza A, Shiva S (2020) Mitochondrial electron transport chain: Oxidative phosphorylation, oxidant production, and methods of measurement. Redox Biol 37:101674. - »Bioblast link«
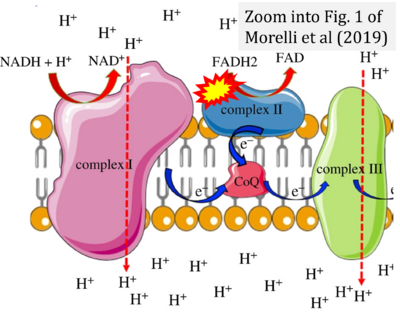
- r Morelli AM, Ravera S, Calzia D, Panfoli I (2019) An update of the chemiosmotic theory as suggested by possible proton currents inside the coupling membrane. Open Biol 9:180221. - »Bioblast link«
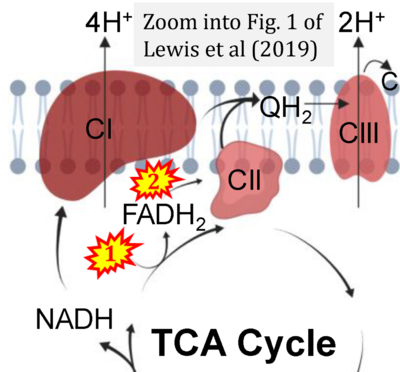
- s Lewis MT, Kasper JD, Bazil JN, Frisbee JC, Wiseman RW (2019) Quantification of mitochondrial oxidative phosphorylation in metabolic disease: application to Type 2 diabetes. Int J Mol Sci 20:5271. - »Bioblast link«
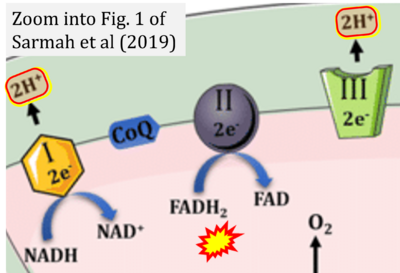
- t Sarmah D, Kaur H, Saraf J, Vats K, Pravalika K, Wanve M, Kalia K, Borah A, Kumar A, Wang X, Yavagal DR, Dave KR, Bhattacharya P (2019) Mitochondrial dysfunction in stroke: implications of stem cell therapy. Transl Stroke Res doi: 10.1007/s12975-018-0642-y - »Bioblast link«
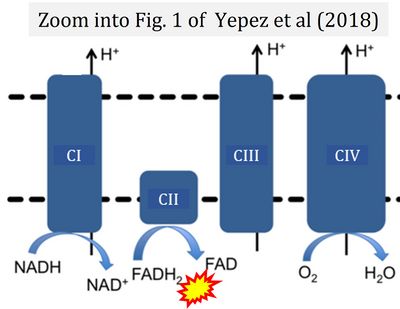
- u Yépez VA, Kremer LS, Iuso A, Gusic M, Kopajtich R, Koňaříková E, Nadel A, Wachutka L, Prokisch H, Gagneur J (2018) OCR-Stats: Robust estimation and statistical testing of mitochondrial respiration activities using Seahorse XF Analyzer. PLOS ONE 13:e0199938. - »Bioblast link«
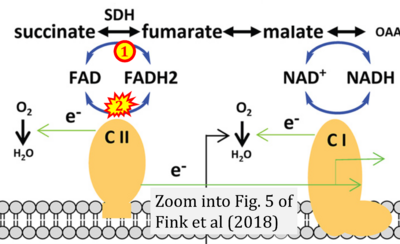
- v Fink BD, Bai F, Yu L, Sheldon RD, Sharma A, Taylor EB, Sivitz WI (2018) Oxaloacetic acid mediates ADP-dependent inhibition of mitochondrial complex II-driven respiration. J Biol Chem 293:19932-41. - »Bioblast link«
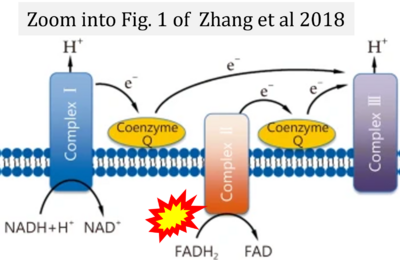
- w Zhang H, Feng YW, Yao YM (2018) Potential therapy strategy: targeting mitochondrial dysfunction in sepsis. Mil Med Res 5:41. - »Bioblast link«
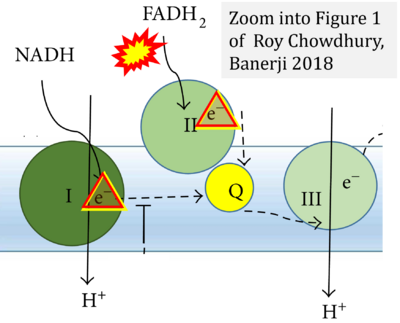
- x Roy Chowdhury S, Banerji V (2018) Targeting mitochondrial bioenergetics as a therapeutic strategy for chronic lymphocytic leukemia. Oxid Med Cell Longev 2018:2426712. - »Bioblast link«
- y de Villiers D, Potgieter M, Ambele MA, Adam L, Durandt C, Pepper MS (2018) The role of reactive oxygen species in adipogenic differentiation. Adv Exp Med Biol 1083:125-144. - »Bioblast link«
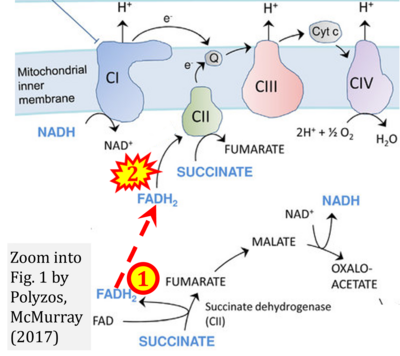
- z Polyzos AA, McMurray CT (2017) The chicken or the egg: mitochondrial dysfunction as a cause or consequence of toxicity in Huntington's disease. Mech Ageing Dev 161:181-97. - »Bioblast link«
- α Jones PM, Bennett MJ (2017) Chapter 4 - Disorders of mitochondrial fatty acid β-oxidation. Elsevier In: Garg U, Smith LD , eds. Biomarkers in inborn errors of metabolism. Clinical aspects and laboratory determination:87-101. - »Bioblast link«
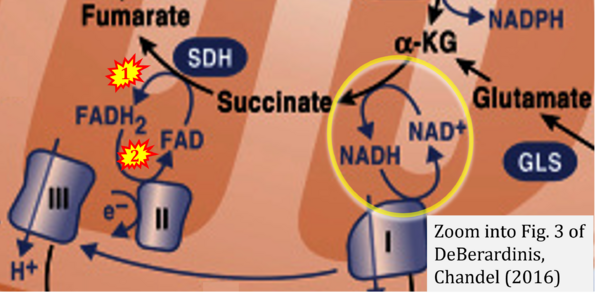
- β DeBerardinis RJ, Chandel NS (2016) Fundamentals of cancer metabolism. Sci Adv 2:e1600200. - »Bioblast link«
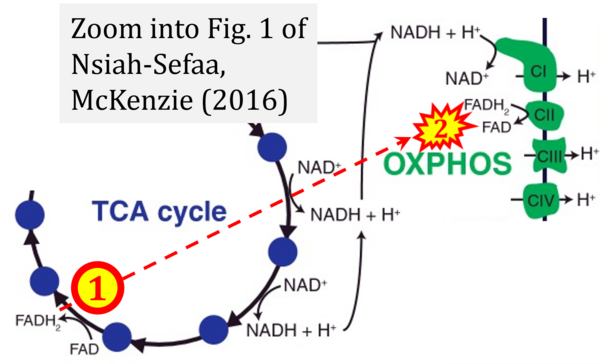
- γ Nsiah-Sefaa A, McKenzie M (2016) Combined defects in oxidative phosphorylation and fatty acid β-oxidation in mitochondrial disease. Biosci Rep 36:e00313. - »Bioblast link«
- δ Prochaska LJ, Cvetkov TL (2013) Mitochondrial electron transport. In: Roberts, G.C.K. (eds) Encyclopedia of Biophysics. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-16712-6_25 - »Bioblast link«
- ε, ζ Fisher-Wellman KH, Neufer PD (2012) Linking mitochondrial bioenergetics to insulin resistance via redox biology. Trends Endocrinol Metab 23:142-53. - »Bioblast link«
- η Benard G, Bellance N, Jose C, Rossignol R (2011) Relationships between mitochondrial dynamics and bioenergetics. In: Lu Bingwei (ed) Mitochondrial dynamics and neurodegeneration. Springer ISBN 978-94-007-1290-4:47-68. - »Bioblast link«
- Θ Nussbaum RL (2005) Mining yeast in silico unearths a golden nugget for mitochondrial biology. J Clin Invest 115:2689-91. - »Bioblast link«
- ί Himms-Hagen J, Harper ME (2001) Physiological role of UCP3 may be export of fatty acids from mitochondria when fatty acid oxidation predominates: an hypothesis. Exp Biol Med (Maywood) 226:78-84. - »Bioblast link«
- κ Sanchez H, Zoll J, Bigard X, Veksler V, Mettauer B, Lampert E, Lonsdorfer J, Ventura-Clapier R (2001) Effect of cyclosporin A and its vehicle on cardiac and skeletal muscle mitochondria: relationship to efficacy of the respiratory chain. Br J Pharmacol 133:781-8. - »Bioblast link«
- λ Brownlee M (2001) Biochemistry and molecular cell biology of diabetic complications. Nature 14:813-20. - »Bioblast link«
- Ref. [34] Arden GB, Ramsey DJ (2015) Diabetic retinopathy and a novel treatment based on the biophysics of rod photoreceptors and dark adaptation. In: Kolb H, Fernandez E, Nelson R, eds. Webvision: The organization of the retina and visual system [Internet]. Salt Lake City (UT): University of Utah Health Sciences Center; 1995-. - »Bioblast link«
Supplement Figure S2 (v2)
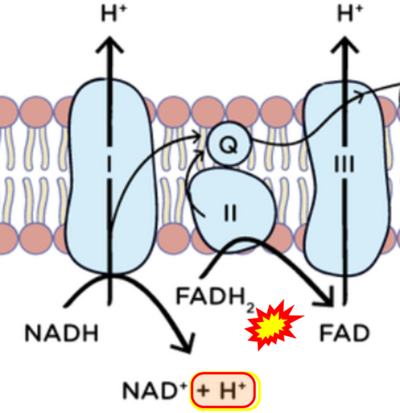
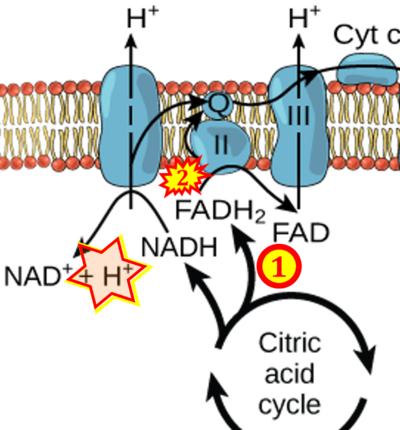
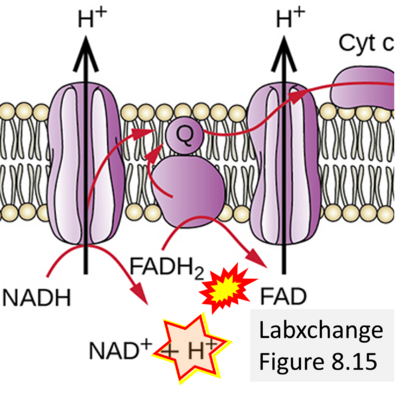
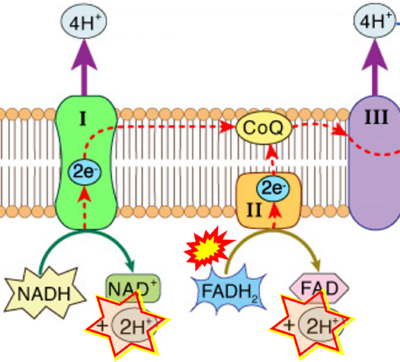
- Figure S2. Complex II ambiguities in graphical representations on FADH2 as a substrate of Complex II in the canonical forward electron transfer (retrieved 2023-03-21 to 2023-04-04)
- Website 1: OpenStax Biology - Fig. 7.10 Oxidative phosphorylation (CC BY 3.0). - OpenStax Biology got it wrong in figures and text. The error is copied without quality assessment and propagated in several links.
- Website 2: Concepts of Biology - 1st Canadian Edition by Charles Molnar and Jane Gair - Fig. 4.19a
- Website 3: LibreTexts Biology Oxidative Phosphorylation - Electron Transport Chain - Figure 7.11.1
- Website 4: lumen Biology for Majors I - Fig. 1
- Website 5: Pharmaguideline
- Website 37: Texas Gateway - Figure 7.11
- Website 38: Concepts of Biology - Charles Molnar and Jane Gair. 4.3 Citric Acid Cycle and Oxidative Phosphorylation. Concepts of Biology - 1st Canadian Edition, BCcampus
- Website 39: -CUNY
- Website 40: - Brain Brooder
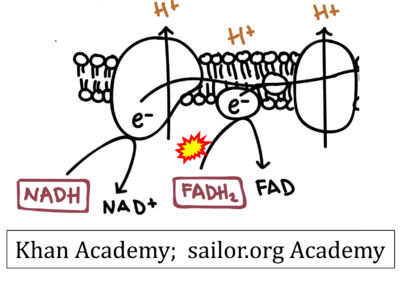
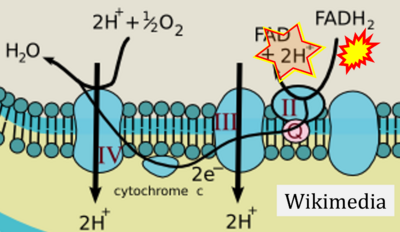
- Website 6: Khan Academy - Image modified from "Oxidative phosphorylation: Figure 1", by OpenStax College, Biology (CC BY 3.0). Figure and text underscore the FADH2-error: "FADH2 .. feeds them (electrons) into the transport chain through complex II."
- Website 7: Saylor Academy
- Website 8: Jack Westin MCAT Courses
- Website 1: OpenStax Biology - Fig. 7.12
- Website 6: Khan Academy - Image modified from "Oxidative phosphorylation: Figure 3," by Openstax College, Biology (CC BY 3.0)
- Website 7: Saylor Academy
- Website 9: expii - Image source: By CNX OpenStax
- Website 37: Texas Gateway - Figure 7.11
- Website 39: -CUNY
- Website 10: Labxchange - Figure 8.15 credit: modification of work by Klaus Hoffmeier
- Website 4: lumen Biology for Majors I - Fig. 3
- Website 9: expii - By OpenStax College CC BY 3.0, via Wikimedia Commons
- Website 11: wikimedia 30148497 - Anatomy & Physiology, Connexions Web site. http://cnx.org/content/col11496/1.6/, 2013-06-19
- Website 12: biologydictionary.net 2018-08-21
- Website 13: Quora
- Website 14: TeachMePhysiology - Fig. 1. 2023-03-13
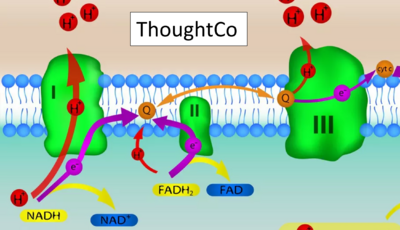
- Website 15: ThoughtCo
- Website 16: toppr
- Website 17: researchtweet
- Website 18: Microbe Notes
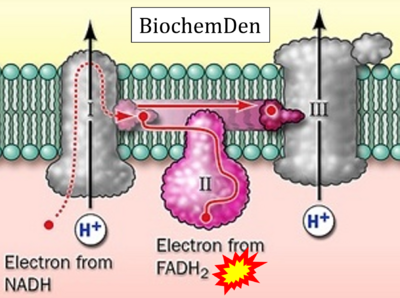
- Website 19: BiochemDen.com
- Website 20: dreamstime
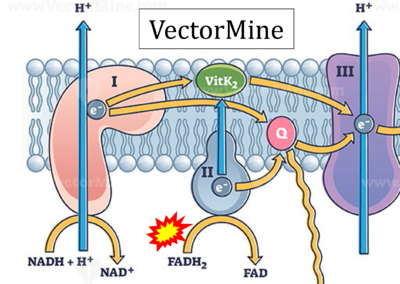
- Website 21: VectorMine
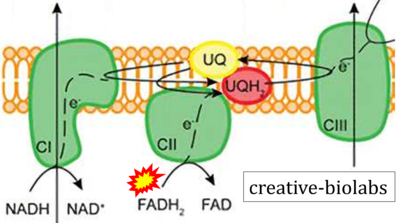
- Website 22: creative-biolabs
- Website 6: Khan Academy
- Website 7: Saylor Academy
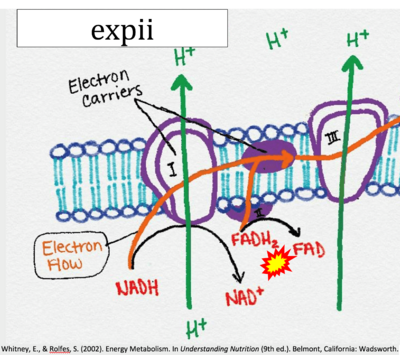
- Website 9: expii - Whitney, Rolfes 2002
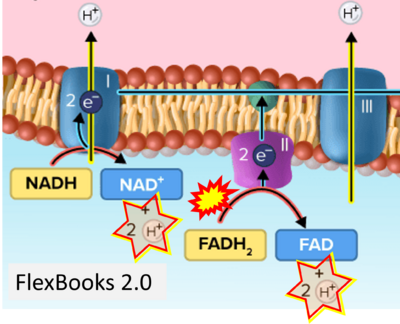
- Website 23: FlexBooks - CK-12 Biology for High School- 2.28 Electron Transport, Figure 2
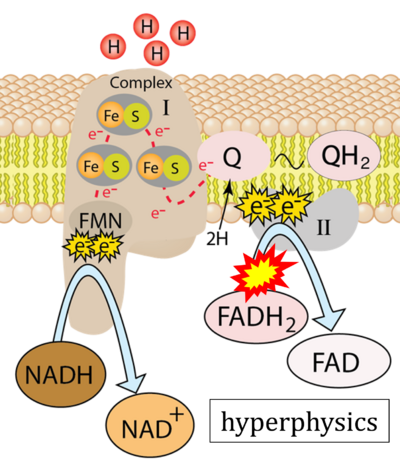
- Website 24: hyperphysics
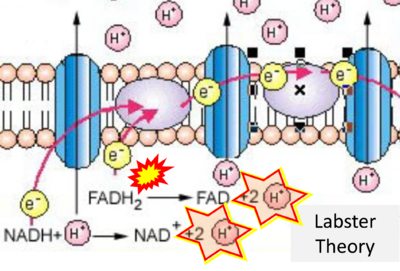
- Website 25: Labster Theory
- Website 26: nau.edu
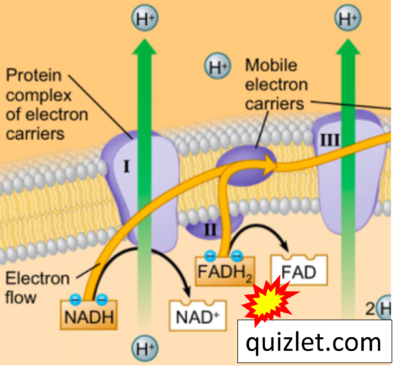
- Website 27: Quizlet
- Website 28: ScienceDirect
- Website 29: ScienceFacts
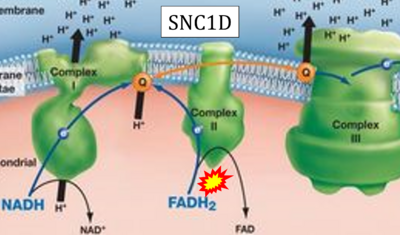
- Website 30: SNC1D - BIOLOGY LESSON PLAN BLOG
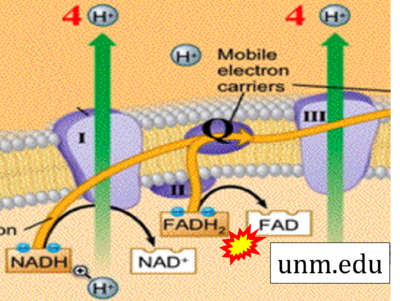
- Website 31: unm.edu
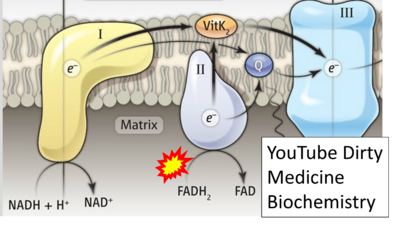
- Website 33: YouTube Dirty Medicine Biochemistry - Uploaded 2019-07-18
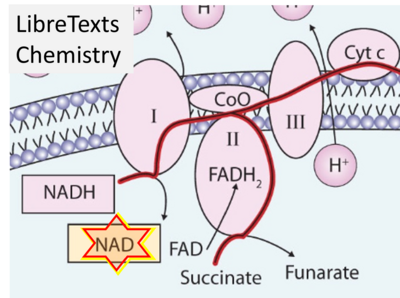
- Website 34: - LibreTexts Chemistry - The Citric Acid Cycle and Electron Transport – Fig. 12.4.3
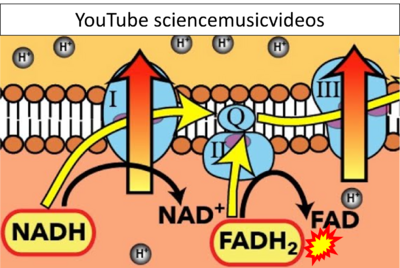
- Website 35: YouTube sciencemusicvideos - Uploaded 2014-08-19
- Website 15: ThoughtCo - extender01 / iStock / Getty Images Plus
- Website 17: dreamstime
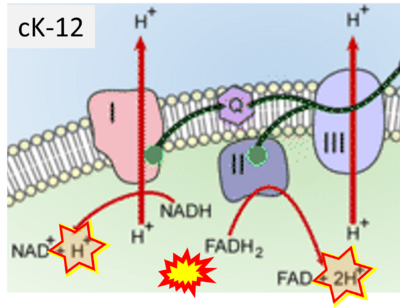
- Website 36: cK-12
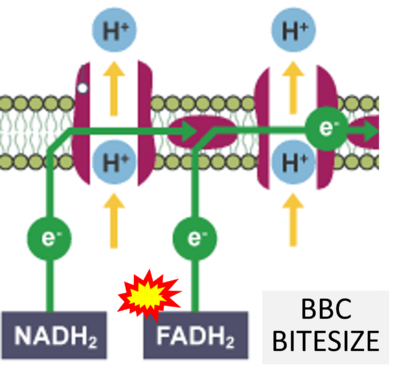
- Website 41: - BBC BITESIZE cK-12
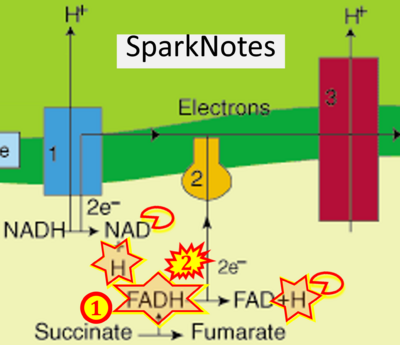
- Website 42: - SparkNotes
- Website 9: expii - Image source: By Gabi Slizewska